
Sevabertinib Approved in China for HER2-Mutant NSCLC: A New Oral Targeted Therapy Option
On April 15, Bayer announced that its innovative targeted therapy Hyrnuo® (sevabertinib, BAY 2927088) has been officially approved by the National Medical Products Administration.
✨ This milestone marks sevabertinib approved in China for HER2-mutant NSCLC, for the treatment of adult patients with unresectable locally advanced or metastatic non-small cell lung cancer (NSCLC) harboring HER2 (ERBB2) activating mutations who have received at least one prior systemic therapy.

The approval of sevabertinib in China for HER2-mutant NSCLC marks a significant milestone, signaling the transition toward oral targeted therapy options in this molecularly defined patient population and improving treatment accessibility.
This article by DengYueMed provides a comprehensive overview of the sevabertinib approved in China for HER2-mutant NSCLC, including its mechanism of action, key clinical data from the SOHO-01 study, safety profile, and clinical positioning, alongside insights into the evolving treatment landscape of HER2-mutant NSCLC.
High Disease Burden: Unmet Needs in HER2-Mutant NSCLC
According to the latest global cancer burden data released by the World Health Organization and International Agency for Research on Cancer (GLOBOCAN 2022), lung cancer has once again become the most commonly diagnosed cancer worldwide: 📊
- Approximately 2.48 million new cases globally, accounting for 12.4% of all cancers
- Around 1.06 million new cases annually in China
- Lung cancer remains the leading cause of cancer-related death
Non-small cell lung cancer (NSCLC) accounts for over 85% of all lung cancer cases.
Among them, HER2 mutations are identified in approximately 2%–4% of advanced NSCLC cases, with a higher prevalence observed in Asian populations. In China, the incidence of HER2-mutant NSCLC is approximately 4.3%.
These mutations, including HER2 exon 20 insertions and point mutations, represent a clearly defined molecular subtype.
😢 However, treatment options for HER2-mutant NSCLC have historically been limited, primarily relying on chemotherapy and antibody-drug conjugates (ADCs), with ongoing challenges in efficacy and tolerability.
For a broader overview of available therapies, see our Complete Lung Cancer Drugs List (2026 Update), which covers current treatment options across different NSCLC subtypes.👇
The emergence of sevabertinib approved in China for HER2-mutant NSCLC addresses a critical unmet need in this setting.
Hyrnuo® (Sevabertinib): Mechanism of Action and Drug Characteristics
Hyrnuo® (sevabertinib) is a next-generation oral tyrosine kinase inhibitor (TKI) developed by Bayer, specifically designed to target aberrant HER2 (ERBB2) signaling pathways in HER2-mutant NSCLC.

From a mechanistic perspective, HER2 activating mutations—especially exon 20 insertions—drive constitutive activation of downstream pathways such as PI3K/AKT and MAPK, promoting tumor cell proliferation and survival.
Sevabertinib is designed to overcome these challenges, with key features including:
1️⃣ Potent inhibition of HER2 mutations
Active against both HER2 exon 20 insertions and point mutations, covering the most common oncogenic drivers in HER2-mutant NSCLC
2️⃣ Reversible ATP-competitive inhibition
Blocks aberrant kinase signaling by targeting the intracellular kinase domain
3️⃣ Selective activity profile
Demonstrates activity against mutant EGFR with improved selectivity compared with wild-type receptors, potentially reducing off-target toxicity
4️⃣ Oral administration for improved adherence
Administered twice daily, supporting long-term disease management
Compared with intravenous ADC therapies, oral TKI treatment options such as sevabertinib offer greater flexibility and convenience, particularly in long-term treatment settings or resource-limited environments.
Notably, sevabertinib has also received accelerated approval from the U.S. Food and Drug Administration, further supporting the global relevance of sevabertinib approved in China for HER2-mutant NSCLC.
Key Clinical Evidence: SOHO-01 Trial Results
The approval of sevabertinib approved in China for HER2-mutant NSCLC is based on results from the multicenter, open-label phase I/II SOHO-01 trial.
Study Design
- Open-label, multicenter study
- A total of 209 patients with HER2-mutant advanced NSCLC (as of June 27, 2025)
- Three cohorts:
- Cohort D: previously treated, no prior HER2-targeted therapy
- Cohort E: prior HER2 ADC therapy
- Cohort F: treatment-naïve patients
Efficacy Outcomes
Objective Response Rate (ORR):
- 64% (Cohort D)
- 38% (Cohort E)
- 71% (Cohort F, exploratory first-line population)
Median Duration of Response (DoR):
- 9.2 months (D)
- 8.5 months (E)
- 11.0 months (F)
Median Progression-Free Survival (PFS):
- 8.3 months (D)
- 5.5 months (E)
- Not reached (F)
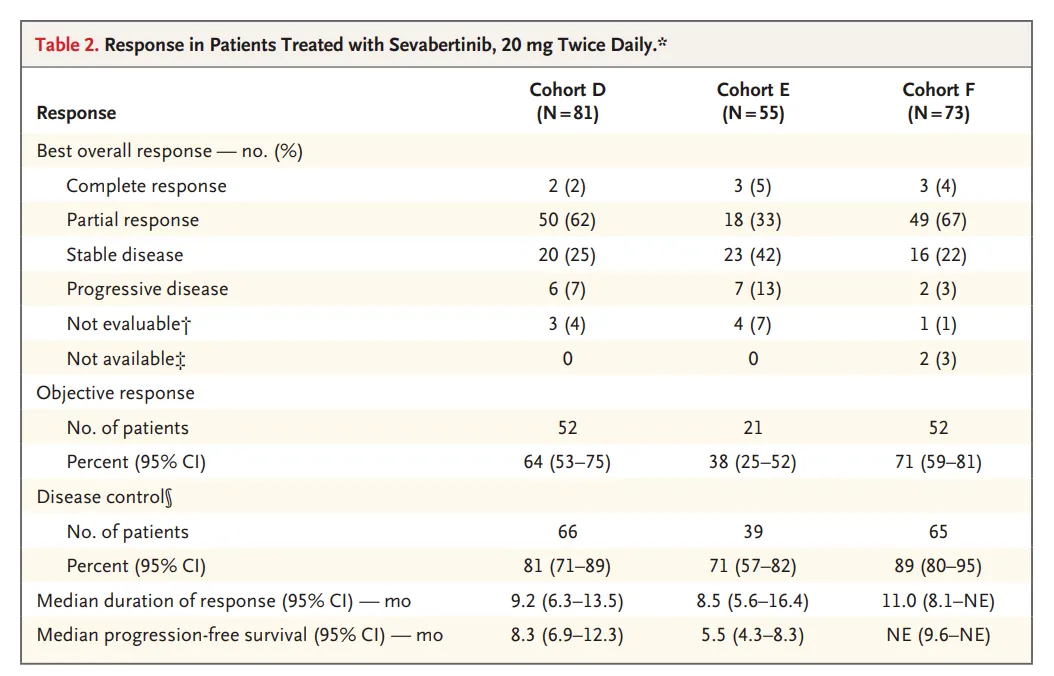
These results demonstrate that sevabertinib in HER2-mutant NSCLC provides clinically meaningful activity, including in patients previously treated with ADC therapies.
Safety and Tolerability
Based on pooled safety data from the SOHO-01 trial:
- Grade ≥3 treatment-related adverse events (TRAEs): ~31%
- Most common adverse event: diarrhea (primarily grade 1–2; grade 3: ~14%)
- Treatment discontinuation rate: ~3%
- No interstitial lung disease (ILD) or treatment-related pneumonitis signals observed during follow-up
Overall, the safety profile of sevabertinib is consistent with that of other TKIs and is considered manageable.
Ongoing Development: SOHO-02 Phase III Trial
The global phase III SOHO-02 trial is currently underway to evaluate sevabertinib as a first-line treatment for HER2-mutant NSCLC.
If confirmed, sevabertinib approved in China for HER2-mutant NSCLC may expand into earlier lines of therapy, potentially reshaping the treatment paradigm and enabling differentiation from ADCs and immunotherapy strategies.
Industry Impact: Advancing Oral Targeted Therapy in HER2-Mutant NSCLC
The approval of Hyrnuo represents more than a single drug launch—it reflects a broader shift in the treatment landscape of HER2-mutant NSCLC.
Historically, despite HER2 being a well-established target in breast and gastric cancers, its role in lung cancer has been more challenging due to mutation heterogeneity and limited therapeutic options.
The introduction of sevabertinib approved in China for HER2-mutant NSCLC brings several key advancements: 🔽
- Filling the gap in oral TKI therapy
Providing a convenient oral alternative to chemotherapy and ADCs - Optimizing treatment sequencing
Offering an additional option before or after ADC therapy - Driving precision medicine adoption
Encouraging broader HER2 testing and molecular profiling - Improving patient accessibility
Oral administration enhances convenience and long-term adherence - Accelerating global innovation
Promoting further drug development and competition in this space
HER2-mutant NSCLC is transitioning from a setting with limited options to one with multiple targeted therapy strategies, including oral TKIs, ADCs, and combination approaches.
Conclusion
The approval of sevabertinib in China for HER2-mutant NSCLC marks a meaningful step forward in precision oncology.
As ongoing studies such as the SOHO-02 trial continue to generate data, sevabertinib approved in China for HER2-mutant NSCLC may further expand its role across treatment lines and clinical settings.
🚀 At the same time, global drug accessibility, supply stability, and information transparency remain critical. China pharmaceutical wholesaler DengYueMed will continue to monitor advancements in HER2-mutant NSCLC and support global pharmaceutical connectivity, contributing to improved access to innovative cancer therapies.
FAQ about Sevabertinib Approved in China for HER2-Mutant NSCLC
What is sevabertinib and how does it work in HER2-mutant NSCLC?
Sevabertinib is an oral tyrosine kinase inhibitor (TKI) that targets HER2 (ERBB2) activating mutations. It works by inhibiting abnormal signaling pathways such as PI3K/AKT and MAPK, which drive tumor growth in HER2-mutant NSCLC.
Who is eligible for sevabertinib treatment in China?
Sevabertinib is approved in China for adult patients with unresectable or metastatic HER2-mutant NSCLC who have received at least one prior systemic therapy. Patients must have confirmed HER2 (ERBB2) activating mutations.
What clinical evidence supports sevabertinib in HER2-mutant NSCLC?
The approval is based on the phase I/II SOHO-01 trial, which demonstrated objective response rates of up to 64% in previously treated patients. The study also showed durable responses and manageable safety.
What are the advantages of sevabertinib as an oral targeted therapy?
As an oral TKI, sevabertinib offers greater convenience compared with intravenous therapies such as ADCs. It also provides a new treatment option for patients with limited choices, including those previously treated with HER2-targeted therapies.



