Monjuvi (Tafasitamab-Cxix) – Lymphoma|HongKong DengYue Medicine
- Generic Name/Brand Name: Tafasitamab-cxix / Minjuvi®
- Indications: R/R DLBCL
- Dosage Form: Injection
- Specification: 200 mg, 10 mL × 1 vial/box
Tafasitamab-Cxix Application Scope
- Diffuse Large B-cell Lymphoma (DLBCL): Approved for relapsed/refractory (R/R) DLBCL in adults who are not eligible for autologous stem cell transplant (ASCT), in combination with lenalidomide.
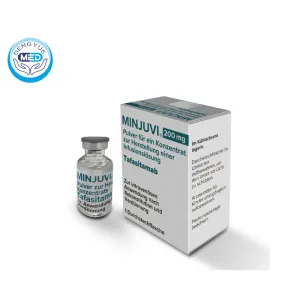
monjuvi tafasitamab-cxix
Tafasitamab-Cxix Characteristics
- Ingredients: Recombinant humanized monoclonal antibody (anti-CD19) with Fc-modification for enhanced immune cell engagement .
- Properties: – Molecular Weight: ~150 kDa (IgG1κ antibody)
– Appearance: Clear to opalescent, colorless to slightly yellow solution . - Specification: Single-dose vials: 200 mg/10 mL (20 mg/mL) .
- Packaging Specification: Carton containing 1 vial .
- Storage: Refrigerate at 2–8°C; protect from light. Do not freeze or shake .
- Expiry Date: Check vial label (typically 24 months from manufacture if unopened) .
- Executive Standard: Manufactured under FDA (U.S.) and EMA (EU) regulations .
- Approval Number: – U.S. FDA Approval: July 2020 (accelerated approval)
– EMA Approval: January 2021 (conditional marketing authorization) . - Date of Revision: Latest updates as of 2023 (post-marketing studies ongoing) .
- Manufacturer: MorphoSys AG (developed in collaboration with Incyte Corporation) .
Guidelines For The Use Of Tafasitamab-Cxix
Dosage and Administration: – Loading Phase: 12 mg/kg IV on Days 1, 4, 8, 15, and 22 of Cycle 1, then weekly in Cycle 2 .
– Maintenance: 12 mg/kg IV on Days 1 and 15 of each subsequent cycle (28-day cycles) until disease progression or unacceptable toxicity .
– Premedication: Administer corticosteroids, antihistamines, and antipyretics to reduce infusion-related reactions .
Adverse Reactions: Most common (≥20%): Neutropenia, fatigue, anemia, diarrhea, thrombocytopenia, cough, fever, peripheral edema .
Serious risks: Infections (e.g., pneumonia), severe infusion-related reactions, tumor lysis syndrome .
Contraindications: Hypersensitivity to tafasitamab or excipients .
Precautions: – Monitor for infections, cytopenias, and infusion-related reactions .
– Vaccination: Avoid live vaccines during treatment .
– Pregnancy/Breastfeeding: Not recommended (potential fetal harm; unknown excretion in milk) .
Tafasitamab-Cxix Interactions
- Drug Interactions: – Lenalidomide: Synergistic effect but may increase myelosuppression (monitor blood counts) .
– Live Vaccines: Contraindicated due to immunosuppression .
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.





Reviews
There are no reviews yet.