
Belantamab Mafodotin Approved in China for Multiple Myeloma: First-in-Class BCMA ADC Marks New Milestone
On April 17, China’s National Medical Products Administration (NMPA) approved belantamab mafodotin in combination with bortezomib and dexamethasone for the treatment of adult patients with relapsed or refractory multiple myeloma who have received at least one prior line of therapy.

This milestone marks belantamab mafodotin approved in China for multiple myeloma, bringing the world’s first approved BCMA-targeted antibody–drug conjugate (ADC) into the Chinese market.
🚀 It also highlights renewed clinical validation of the ADC approach amid the rapid evolution of CAR-T therapies and bispecific antibodies.
China-based pharmaceutical distributor DengYueMed provides an industry and clinical perspective on belantamab mafodotin approved in China for multiple myeloma, focusing on its latest developments and clinical significance.
Multiple Myeloma: Disease Background and Treatment Challenges
Multiple myeloma is a hematologic malignancy characterized by the abnormal proliferation of plasma cells.
Current treatment paradigms are built on three major drug classes: proteasome inhibitors, immunomodulatory drugs (IMiDs), and anti-CD38 monoclonal antibodies.
Despite these advances, clinical challenges remain significant. Most patients eventually relapse, and treatment efficacy declines with successive lines of therapy, accompanied by increasing drug resistance. 😢
Unmet medical needs are particularly pronounced in: 🔽
- Lenalidomide-refractory patients
- High-risk cytogenetic subgroups
- Patients with multiple prior lines of therapy
In this context, B-cell maturation antigen (BCMA) has emerged as one of the most validated therapeutic targets due to its high and selective expression on malignant plasma cells.
The approval of belantamab mafodotin approved in China for multiple myeloma further reinforces the importance of BCMA-targeted strategies.
Belantamab Mafodotin: Mechanism of Action and ADC Design
Blenrep (belantamab mafodotin) is a BCMA-directed ADC designed to deliver cytotoxic payloads directly to tumor cells.
It consists of a humanized anti-BCMA monoclonal antibody conjugated via a non-cleavable linker to monomethyl auristatin F (MMAF), a microtubule inhibitor.
🎯 Its anti-tumor activity involves multiple mechanisms:
- Binding to BCMA on myeloma cells followed by internalization and intracellular release of the cytotoxic agent
- Induction of antibody-dependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP)
- Potential induction of immunogenic cell death (ICD), enhancing anti-tumor immune responses
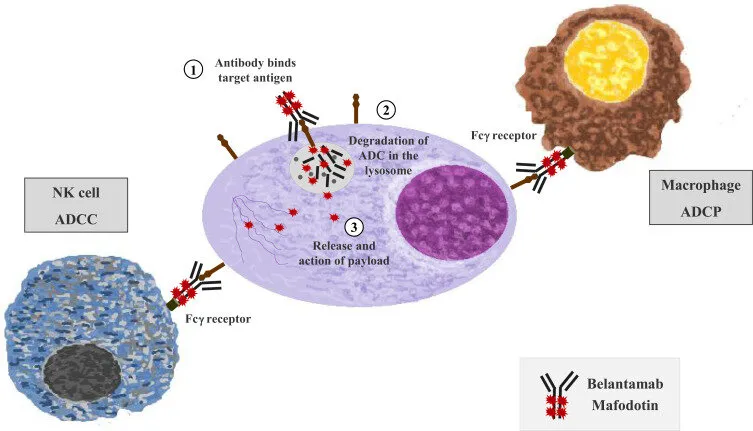
This dual “targeted + immune-mediated” mechanism supports its use in combination regimens and underpins the clinical value of belantamab mafodotin approved in China for multiple myeloma.
Clinical Evidence Evolution: From Setback to Re-establishment
The development trajectory of blenrep reflects a typical pattern of initial success, subsequent setback, and eventual revalidation.
The phase II DREAMM-2 trial supported its accelerated approval in 2020. However, the phase III DREAMM-3 study failed to meet its primary endpoint, leading to market withdrawal in the US and EU and highlighting the limitations of ADC monotherapy.
A turning point came with the phase III DREAMM-7 trial, which evaluated belantamab mafodotin combined with bortezomib and dexamethasone (BVd) versus daratumumab plus bortezomib and dexamethasone (DVd) in relapsed/refractory patients.
Key results demonstrated: 🔽
- Significantly improved progression-free survival (PFS): 36.6 vs 13.4 months (HR 0.41)
- 42% reduction in risk of death (HR 0.58)
- Higher overall response rate (ORR) and deeper responses
👉 These findings were published in the New England Journal of Medicine and presented at American Society of Clinical Oncology and American Society of Hematology annual meetings.
Together, they demonstrate that ADCs can achieve substantial clinical benefit when used in combination strategies, further supporting belantamab mafodotin approved in China for multiple myeloma.
Chinese Subgroup Analysis: Consistent and Enhanced Benefit
In subgroup analyses presented at the European Hematology Association Congress, including Chinese patients, the efficacy of blenrep was further validated.
With a median follow-up of 26.5 months: 🔽
- Median PFS: not reached vs 8.4 months (HR 0.24)
- Median OS: not reached vs 38.4 months (HR 0.29)
- ORR: 81.3% vs 67.5%
These results suggest not only consistent benefit but also potentially greater risk reduction in Chinese patients, reinforcing the clinical relevance of belantamab mafodotin approved in China for multiple myeloma in real-world practice.
Safety Profile and Clinical Management
The most notable adverse events associated with blenrep 100mg are corneal toxicities, including keratopathy, blurred vision, and dry eye.
These events are generally manageable with appropriate monitoring and dose adjustments.
Key management strategies include: 👇
- Regular ophthalmologic assessments
- Dose modifications or treatment delays when necessary
- Supportive care such as artificial tears
With proper management, most adverse events are reversible, and discontinuation rates due to ocular toxicity remain relatively low (≤9%).

BCMA Landscape: Increasingly Competitive Modalities
The BCMA-targeted therapeutic landscape is rapidly evolving, with multiple modalities coexisting in China:
- CAR-T therapies: ciltacabtagene autoleucel, zevorcabtagene autoleucel
- Bispecific antibodies: elranatamab, teclistamab
Each modality presents distinct characteristics.
1️⃣ CAR-T therapies offer deep and durable responses but are limited by cost and logistical complexity.
2️⃣ Bispecific antibodies provide off-the-shelf convenience but require careful management of immune-related toxicities.
3️⃣ ADCs, including belantamab mafodotin, offer a balanced profile in terms of efficacy, safety, and accessibility. For a broader overview of the evolving ADC landscape, see our article: 👇
As a result, treatment strategies are shifting from a single “best option” toward a more integrated, multi-modality approach.
Conclusion: From Validation to Long-Term Competition
The milestone of belantamab mafodotin approved in China for multiple myeloma represents more than a regulatory approval—it signifies a critical turning point for ADCs in hematologic malignancies.
The transition from monotherapy limitations to combination-based success highlights a broader evolution in treatment strategy.
Looking ahead, several trends are likely to shape the field:
- Combination regimens moving into earlier lines of therapy
- Increasing use of biomarker-driven patient stratification
- Competition expanding beyond efficacy to include safety, accessibility, and cost
At the same time, next-generation BCMA-targeted therapies—including improved ADCs, dual-target bispecific antibodies, and off-the-shelf CAR-T platforms—are rapidly advancing.
As a China-based pharmaceutical distributor, DengYueMed continues to monitor innovations such as blenrep and, within a compliant framework, supports supply and distribution systems to enhance treatment accessibility for multiple myeloma and other therapeutic areas.
FAQ about Belantamab Mafodotin Approved in China for Multiple Myeloma
What is Belantamab Mafodotin and how does it work?
Blenrep (belantamab mafodotin) is a BCMA-targeted antibody–drug conjugate (ADC) that delivers a cytotoxic payload directly to myeloma cells. It binds to BCMA on tumor cells, is internalized, and releases a microtubule inhibitor to induce cell death.
Who can receive Belantamab Mafodotin in China?
Belantamab mafodotin is approved in China for adult patients with relapsed or refractory multiple myeloma who have received at least one prior line of therapy. It is used in combination with bortezomib and dexamethasone. This provides an additional treatment option for patients with limited alternatives.
What are the key clinical benefits of Belantamab Mafodotin?
Clinical data from the DREAMM-7 study showed significant improvements in progression-free survival and overall survival compared with standard regimens. The combination therapy reduced the risk of disease progression or death and achieved higher response rates.
What are the main safety considerations?
The most notable side effects are eye-related events such as keratopathy and blurred vision. These are generally manageable with regular monitoring, dose adjustments, and supportive care. Most adverse events are reversible when properly managed.



