Latest Advances in Thalassemia Gene Therapy: How CRISPR and Gene Addition Therapies Enable TDT Patients to Become Transfusion-Independent
For patients with transfusion-dependent thalassemia (TDT), long-term blood transfusions have traditionally been the cornerstone of treatment, but they also bring a substantial medical burden.
From early childhood, patients typically require transfusions every 2 to 5 weeks, along with lifelong iron chelation therapy to prevent irreversible damage caused by iron overload in the heart, liver, and endocrine system. 😢
This treatment regimen not only imposes a continuous physical burden but also places significant financial pressure on families. Recent advances in thalassemia gene therapy offer new hope for these patients.
As a global pharmaceutical distributor connecting innovative therapies with clinical needs, DengYueMed continuously monitors the development of cutting-edge technologies such as gene therapy.
✨ In recent years, with the maturation of gene addition and CRISPR gene editing technologies, thalassemia treatment is undergoing a profound shift from “long-term maintenance” to “functional cure.”
1. Thalassemia: Unmet Clinical Needs
Thalassemia is a group of hereditary hemolytic anemias caused by mutations or deletions in globin genes, primarily classified into alpha (α) and beta (β) types.
According to global data on thalassemia prevalence from the World Health Organization, millions of people are affected worldwide. Approximately 5% of the global population carries related mutations, with southern China being a high-prevalence region, where carriers exceed 30 million and severe patient numbers are substantial.
Due to insufficient beta-globin synthesis, patients have abnormal hemoglobin structure, leading to shortened red blood cell lifespan, increased ineffective erythropoiesis, and progressive complications such as bone marrow expansion, skeletal deformities, and multi-organ damage.
Limitations of Traditional Treatment
Currently, TDT treatment relies on regular transfusions to maintain hemoglobin levels, typically around 90–105 g/L.
However, the human body lacks an active iron excretion mechanism. Long-term transfusions inevitably lead to iron accumulation, damaging critical organs such as the heart, liver, and pancreas. Consequently, patients must undergo lifelong iron chelation therapy.
Allogeneic hematopoietic stem cell transplantation (HSCT) can achieve a cure, but it is limited by donor availability, transplant-related mortality, and graft-versus-host disease (GVHD) risk. As a result, less than 10% of patients truly benefit.
This reality leaves the vast majority of patients in a “manageable but not curable” state.
2. Emergence of Thalassemia Gene Therapy
Gene therapy aims to repair or replace defective genes, restoring normal function to the patient’s hematopoietic system.
The core process involves harvesting autologous hematopoietic stem cells, genetically modifying them ex vivo, and reinfusing them into the patient, achieving long-term or potentially lifelong therapeutic effects.
This strategy also avoids dependence on donors and immune rejection, making gene therapy a highly promising treatment approach for transfusion-dependent thalassemia (TDT).
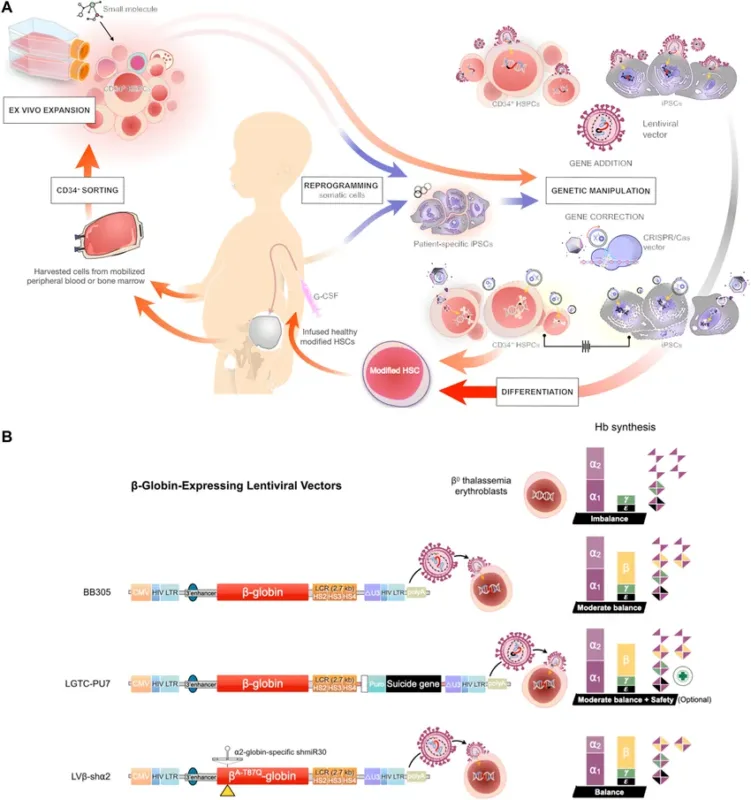
thalassemia gene therapy process hematopoietic stem cells
3. Gene Therapy Approaches
Gene Addition: Compensating Beta-Globin Deficiency
Gene addition therapy uses lentiviral vectors to deliver functional beta-globin genes into patient hematopoietic stem cells.
Since beta-globin expression is tightly regulated, vectors typically include key regulatory elements such as the locus control region (LCR) to ensure high and stable expression in erythroid cells.
Lentiviral vectors integrate into the host genome and maintain expression as stem cells differentiate, enabling long-term production of functional hemoglobin.
However, expression levels are influenced by integration sites and vector copy numbers, making manufacturing optimization essential for therapeutic efficacy.
CRISPR Gene Editing: Reactivating Fetal Hemoglobin (HbF)
The CRISPR/Cas9 gene editing strategy does not directly repair the beta-globin gene. Instead, it modulates gene expression networks to achieve therapeutic effect.
Research shows that BCL11A is a key transcription factor that suppresses fetal hemoglobin (HbF) expression. By editing its erythroid-specific enhancer, BCL11A expression in red blood cell precursors can be selectively reduced, reactivating gamma-globin (γ-globin) synthesis.
HbF has normal oxygen-carrying capacity and is unaffected by beta-globin mutations, making it a functional substitute for adult hemoglobin.
This “indirect repair” mechanism is central to CRISPR-based thalassemia gene therapy.
Comparing the Two Approaches
Overall, both approaches have achieved high rates of transfusion independence in clinical practice. Gene addition therapy benefits from more mature long-term follow-up data, while CRISPR therapy avoids exogenous gene integration and has broad potential applicability.
It is important to note that current major risks for both approaches are not from the gene modification itself but from pre-treatment myeloablative conditioning, which remains a key area for future optimization.
The ultimate goal of gene therapy is to restore normal red blood cell structure and function.
4. Global and Chinese Thalassemia Gene Therapy Clinical Progress
Zynteglo™: Long-Term Validation of Gene Addition Therapy
Mechanism: Zynteglo delivers a beta-globin gene (HbAT87Q) into patient CD34+ hematopoietic stem cells via lentiviral vector, restoring red blood cell oxygen-carrying capacity.
- Long-term follow-up (median ~5.9 years, maximum >10 years) in Phase III thalassemia gene therapy clinical trials showed ~90% of patients achieved transfusion independence.
- Hemoglobin levels maintained around 11 g/dL; markers of ineffective erythropoiesis improved significantly.
- Over 70% of patients discontinued iron chelation therapy later, without increases in liver iron concentration.
- Safety data: No insertional oncogenesis or abnormal clonal expansion observed, demonstrating a stable risk profile.
Casgevy™: CRISPR Therapy Milestone
Mechanism: Casgevy uses CRISPR/Cas9 to target the BCL11A erythroid enhancer, reducing its expression to derepress γ-globin synthesis, thereby increasing HbF levels and functionally replacing adult hemoglobin.
Key Clinical Studies:
- ~98% of TDT patients achieved at least 12 months of transfusion independence, with an average duration over 40 months.
- Some patients stopped transfusions within months post-treatment, maintaining stable hemoglobin levels.
- Pediatric data: Most children achieved transfusion independence rapidly.
- Safety: Rare severe adverse events were linked to myeloablative preconditioning, indicating the need for risk optimization.
Hemo-Cel: Breakthrough in China
Mechanism: Hemo-Cel uses a lentiviral vector to deliver functional beta-globin genes, restoring normal differentiation of hematopoietic stem cells for continuous hemoglobin production.
Key Clinical Studies:
- All evaluable patients achieved transfusion independence, with median follow-up >34 months, maximum >5 years.
- Hemoglobin levels remained stable; iron load decreased; no virus-related safety issues or abnormal clonal expansion observed.
- Demonstrates that domestically developed Chinese gene therapy products have international competitiveness and provide regional patient access.
5. Comparative Clinical Evidence
Currently, multiple thalassemia gene therapy clinical studies have reached critical stages, and DengYueMed continuously tracks and updates the latest developments in this field.
To illustrate the differences between gene addition and CRISPR therapies, the following horizontal analysis compares technology pathways and key clinical data:
| Indicator | Zynteglo™ | Casgevy™ (≥12 yrs) | Casgevy™ (5–11 yrs) | Hemo-Cel |
|---|---|---|---|---|
| Technology | Gene addition | Gene editing (BCL11A) | Gene editing (BCL11A) | Gene addition |
| Exogenous gene | Yes | No | No | Yes |
| Transfusion independence | 90.2% (37/41) | 98.2% (55/56) | 100% (6/6) | 100% (5/5) |
| Hb / HbF | Hb≈11.2 g/dL | HbF significantly ↑ | Hb≥9 g/dL | Not reported |
| Duration off transfusion | Up to 10.1 yrs | Avg. 41.4 months | Avg. 19.8 months | Up to 61 months |
| Follow-up | Median 5.9 yrs (max >10 yrs) | Approx. 3–4 yrs | Approx. 2 yrs | Approx. 5 yrs |
Both approaches achieve transfusion independence rates above 90%, marking a new stage of functional cure in TDT treatment.
From the perspective of evidence: Gene addition therapy provides long-term stable efficacy, while gene editing therapy demonstrates rapid short-term efficacy and mechanistic innovation, with long-term data still being accumulated.
6. Real-World Considerations in Clinical Decision-Making
In real-world clinical settings, therapy choice depends not only on efficacy but also on multiple considerations:
1️⃣ Long-term stability: For chronic genetic diseases, sustained efficacy is more important than short-term response.
2️⃣ Individual differences: Including genotype, disease burden, and prior treatment history.
3️⃣Evidence maturity: Some therapies show excellent short-term results, but long-term data are still being collected.
4️⃣ Accessibility and cost: Practical factors that cannot be ignored.
Thus, the two technology pathways are not mutually exclusive but are selected based on patient-specific needs.
7. Future Directions in Thalassemia Gene Therapy: From Technological Breakthrough to Clinical Implementation
Although gene therapy approaches are approaching “curative levels,” widespread application faces key challenges:
- High costs
- Complex manufacturing processes
- Toxicity of myeloablative preconditioning
Future core developments include:
- In vivo gene therapy to simplify procedures
- More precise gene editing techniques to enhance safety
- Milder preconditioning regimens to reduce toxicity
These advances will determine whether thalassemia gene therapy can transition from a “high-end treatment” to a routine clinical option.
Conclusion
As thalassemia gene therapy continues to advance, long-term data from CRISPR and gene addition therapies will further clarify their clinical value.
As a global pharmaceutical distributor, HongKong DengYueMed is committed to promoting global accessibility of innovative therapies. 🚀
With technology maturation, achieving transfusion independence is no longer just a goal—it is increasingly becoming a reality.
FAQ about Thalassemia Gene Therapy
Is there gene therapy for thalassemia?
Yes. Gene therapy for thalassemia exists and has been shown to help patients, especially those with transfusion-dependent thalassemia (TDT), produce functional hemoglobin and reduce or even eliminate the need for regular blood transfusions. Current approaches include gene addition therapy and CRISPR-based gene editing.
What is the latest treatment for thalassemia?
The latest treatments for thalassemia include gene therapy approaches, such as gene addition therapy (e.g., Zynteglo) and CRISPR-based gene editing (e.g., Casgevy), which aim to restore normal hemoglobin production and help patients become transfusion-independent.
Who is eligible for CASGEVY treatment?
CASGEVY treatment is currently approved for patients with transfusion-dependent beta-thalassemia (TDT), typically aged 12 years and older, who require regular blood transfusions to maintain hemoglobin levels. Eligibility may depend on factors such as overall health, prior treatments, and suitability for myeloablative preconditioning before gene therapy.
Can thalassemia be cured permanently?
Currently, thalassemia cannot be permanently cured for all patients, but gene therapy can achieve long-term or potentially lifelong transfusion independence in many cases, effectively functioning as a “functional cure.”