Gene Therapy for Beta Thalassemia: Can CRISPR and Gene Addition Achieve Transfusion Independence?
For patients living with transfusion-dependent beta thalassemia (TDT), lifelong blood transfusions have long been the standard of care.
While effective in maintaining hemoglobin levels, this approach brings a significant burden—requiring frequent transfusions and continuous iron chelation therapy to prevent organ damage. 😢
🚀 Today, gene therapy for beta thalassemia is rapidly transforming this landscape. With the emergence of CRISPR-based gene editing and gene addition therapies, a growing number of patients are achieving transfusion independence, marking a shift toward a functional cure.
As a global pharmaceutical distributor connecting innovative therapies with clinical needs, DengYueMed continuously monitors the development of cutting-edge technologies such as gene therapy.
Understanding Beta Thalassemia and Current Treatment Challenges
Beta thalassemia is a hereditary blood disorder caused by mutations in the beta-globin gene, leading to reduced or absent hemoglobin production.
As a result, patients experience chronic anemia, ineffective erythropoiesis, and a range of long-term complications affecting the heart, liver, and endocrine system.
In severe cases such as TDT, patients typically require blood transfusions every 2–5 weeks. However, because the body cannot actively eliminate excess iron, repeated transfusions inevitably lead to iron overload.
Although hematopoietic stem cell transplantation (HSCT) offers a potential cure, its application is limited by donor availability, transplant-related risks, and complications such as graft-versus-host disease (GVHD).
These limitations highlight the urgent need for innovative approaches such as gene therapy for beta thalassemia.
How Gene Therapy for Beta Thalassemia Works
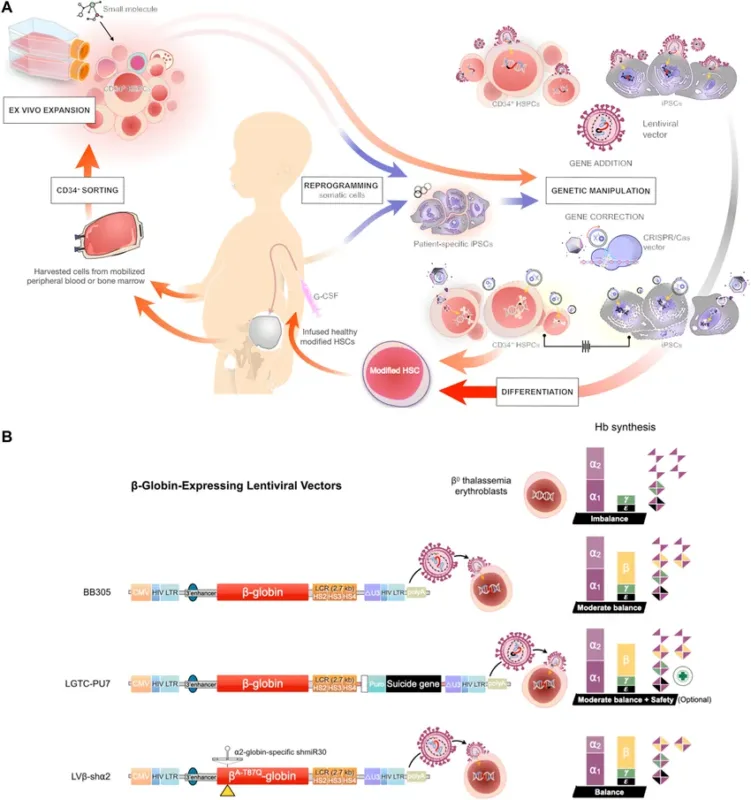
Modern gene therapy for beta thalassemia focuses on correcting or bypassing the underlying genetic defect responsible for abnormal hemoglobin production.
The core process involves:
- Collecting the patient’s own hematopoietic stem cells
- Genetically modifying them ex vivo
- Reinfusing them back into the patient
This approach restores the body’s ability to produce functional hemoglobin while avoiding donor dependency and immune rejection.
thalassemia gene therapy process hematopoietic stem cells
Gene Addition vs CRISPR: Two Key Approaches
Gene Addition: Compensating Beta-Globin Deficiency
Gene addition is one of the most established strategies in gene therapy for beta thalassemia.
This approach uses lentiviral vectors to introduce a functional beta-globin gene into the patient’s stem cells. Once reinfused, these modified cells can continuously produce functional hemoglobin.
👉 Key advantages include:
- Long-term clinical validation
- Stable hemoglobin production
- Sustained therapeutic effect over years
CRISPR Gene Editing: Reactivating Fetal Hemoglobin (HbF)
CRISPR-based therapy represents a major breakthrough in gene therapy for beta thalassemia.
Instead of inserting a new gene, this method edits gene regulation pathways. Specifically, it targets the BCL11A enhancer, reducing its expression and reactivating fetal hemoglobin (HbF).
HbF can effectively compensate for defective adult hemoglobin, restoring oxygen transport capacity.
👉 Key advantages include:
- Broad applicability across patients
- No need for exogenous gene insertion
- Precise genome editing
Comparing the Two Approaches
Overall, both approaches have achieved high rates of transfusion independence in clinical practice. Gene addition therapy benefits from more mature long-term follow-up data, while CRISPR therapy avoids exogenous gene integration and has broad potential applicability.
It is important to note that current major risks for both approaches are not from the gene modification itself but from pre-treatment myeloablative conditioning, which remains a key area for future optimization.
The ultimate goal of gene therapy is to restore normal red blood cell structure and function.
Can Gene Therapy Achieve Transfusion Independence? Clinical Evidence from Beta Thalassemia
Gene Addition Therapy: Long-Term Outcomes
Gene addition remains one of the most established approaches in gene therapy for beta thalassemia, supported by long-term clinical data.
Zynteglo
Mechanism:
Zynteglo delivers a functional beta-globin gene (HbAT87Q) into autologous CD34+ hematopoietic stem cells using a lentiviral vector, enabling sustained production of functional hemoglobin.
Key clinical findings:
- Approximately 90% of patients achieved transfusion independence in Phase III studies
- Median follow-up of ~5.9 years, with some patients exceeding 10 years
- Hemoglobin levels stabilized around ~11 g/dL
- Significant improvement in markers of ineffective erythropoiesis
- Over 70% of patients discontinued iron chelation therapy without increased liver iron concentration
Safety profile:
No evidence of insertional oncogenesis or abnormal clonal expansion has been observed, supporting a stable long-term safety profile.
👉 These results demonstrate the durability and clinical maturity of gene addition strategies in gene therapy for beta thalassemia.
CRISPR-Based Gene Editing: High Response and Rapid Onset
CRISPR-based approaches represent a major advancement in gene therapy for beta thalassemia, particularly in achieving rapid and high rates of response.
Casgevy
Mechanism:
Casgevy uses CRISPR/Cas9 to target the BCL11A erythroid enhancer, reducing its expression and reactivating fetal hemoglobin (HbF), which compensates for defective adult hemoglobin.
Key clinical findings:
- ~98% of patients achieved at least 12 months of transfusion independence
- Average duration of transfusion independence exceeded 40 months
- Some patients discontinued transfusions within months after treatment
- Pediatric patients demonstrated similarly strong responses
Safety considerations:
Severe adverse events were rare and primarily associated with myeloablative preconditioning rather than the gene editing process itself.
👉 These findings highlight the strong efficacy and rapid clinical benefit of CRISPR-based gene therapy for beta thalassemia.
Emerging Gene Therapy Developments
In addition to established therapies, new gene addition platforms are being developed globally, further expanding the therapeutic landscape. See more in our news section.
Hemo-Cel
Mechanism:
Hemo-Cel uses lentiviral-mediated delivery of functional beta-globin genes to restore normal hematopoietic stem cell differentiation and sustained hemoglobin production.
Key clinical findings:
- All evaluable patients achieved transfusion independence
- Median follow-up exceeded 34 months, with some cases beyond 5 years
- Hemoglobin levels remained stable, with reduced iron burden
- No vector-related safety concerns or abnormal clonal expansion observed
👉 These findings highlight the expanding role of China-based research programs in gene therapy development for beta thalassemia.
Comparative Clinical Outcomes
Available clinical data provide a comparative view of outcomes across gene addition and gene editing approaches in gene therapy for beta thalassemia.
| Indicator | Zynteglo™ | Casgevy™ (≥12 yrs) | Casgevy™ (5–11 yrs) | Hemo-Cel |
|---|---|---|---|---|
| Technology | Gene addition | Gene editing (BCL11A) | Gene editing (BCL11A) | Gene addition |
| Exogenous gene | Yes | No | No | Yes |
| Transfusion independence | 90.2% (37/41) | 98.2% (55/56) | 100% (6/6) | 100% (5/5) |
| Hb / HbF | Hb≈11.2 g/dL | HbF significantly ↑ | Hb≥9 g/dL | Not reported |
| Duration off transfusion | Up to 10.1 yrs | Avg. 41.4 months | Avg. 19.8 months | Up to 61 months |
| Follow-up | Median 5.9 yrs (max >10 yrs) | Approx. 3–4 yrs | Approx. 2 yrs | Approx. 5 yrs |
From the perspective of evidence: Gene addition therapy provides long-term stable efficacy, while gene editing therapy demonstrates rapid short-term efficacy and mechanistic innovation, with long-term data still being accumulated.
Safety and Current Challenges
Despite promising outcomes, several challenges remain.
1️⃣ Preconditioning Toxicity
Most gene therapy protocols require myeloablative conditioning, which carries short-term risks.
2️⃣ Cost and Accessibility
High treatment costs limit widespread adoption.
3️⃣ Manufacturing Complexity
Production processes remain technically demanding.
Importantly, current evidence suggests that major risks are primarily associated with preconditioning rather than the gene therapy itself.
Future Directions in Gene Therapy for Beta Thalassemia
Ongoing innovation is expected to further improve outcomes and accessibility.
Key directions include:
- Development of in vivo gene therapy
- More precise gene editing technologies
- Reduced-intensity conditioning regimens
- Expanded global access to advanced therapies
These advances will shape the next phase of gene therapy for beta thalassemia.
Conclusion
The emergence of gene therapy is redefining treatment expectations for beta thalassemia.
Both gene addition and CRISPR-based approaches have demonstrated the ability to achieve high rates of transfusion independence, significantly reducing disease burden.
✨ As clinical experience continues to grow, gene therapy for beta thalassemia is moving closer to becoming a widely accessible and potentially curative treatment option.
As a global pharmaceutical distributor, HongKong DengYueMed is committed to promoting global accessibility of innovative therapies.
FAQ about Gene Therapy for Beta Thalassemia
Is there a gene therapy for beta thalassemia?
Yes. Gene therapy for beta thalassemia is available and can help many patients achieve long-term transfusion independence using approaches like gene addition and CRISPR-based editing.
Is gene therapy 100% safe?
Current data indicate that most risks are related to pre-treatment conditioning, while gene modification itself has shown a favorable safety profile.
What is the latest treatment for beta thalassemia?
The latest treatment for beta thalassemia is gene therapy, including CRISPR-based gene editing and gene addition approaches that target the root genetic cause. These therapies have enabled many patients to achieve long-term transfusion independence.
Can CRISPR cure beta thalassemia?
CRISPR therapies can enable long-term transfusion independence in many patients, functioning as a potential curative approach, although long-term data are still being collected.



