
Drug Treatment of Duchenne Muscular Dystrophy (DMD): Current Therapies, Gene Therapy Advances, and Future Trends
In 1852, French physician Duchenne described boys with progressive muscle weakness whose enlarged calves were replaced by fat and fibrous tissue, leading to death from respiratory or cardiac complications in adolescence or early adulthood. The disease was later named Duchenne Muscular Dystrophy (DMD).
20th-century research further confirmed the presence of myofibrous necrosis, fat deposition, and fibrosis in the skeletal muscles of patients, which could explain the muscle weakness and pseudohypertrophy, but the cause remained unclear.
Until the 1980s, the pathogenic gene was located on the X chromosome Xp21 and cloned—this 79-exon gene encodes dystrophin, a protein that maintains muscle membrane stability, revealing the molecular nature of DMD for the first time.
Thanks to a deeper understanding of disease mechanisms and continuous advances in the drug treatment of Duchenne muscular dystrophy, patient survival has significantly improved, with median life expectancy increasing from the early twenties in previous decades to over 30–40 years in many developed regions today.
Since the discovery of the key pathogenic gene, research on Duchenne muscular dystrophy has continued for more than 40 years. What breakthroughs have been made in diagnosis and treatment? 👩🔬
HongKong DengYue Medicine has compiled and reviewed the current drug treatment of Duchenne Muscular Dystrophy, updated the progress of DMD drug development in China, and is committed to improving global accessibility and supply of these innovative therapies.
Corticosteroids for DMD
It is now clear that Duchenne muscular dystrophy is an X-linked recessive genetic disorder caused by mutations in the dystrophin gene on the X chromosome. It primarily affects boys, with a prevalence of approximately 1 in 3500.
This gene defect leads to the absence of dystrophin, resulting in decreased muscle cell membrane stability and a persistent inflammatory response, ultimately causing muscle fiber necrosis, fatty infiltration, and fibrosis.
Due to their potent anti-inflammatory and immunomodulatory effects, corticosteroids (mainly glucocorticoids) have long been recognized as the standard basic treatment for DMD.
Currently, oral corticosteroids used to treat DMD mainly include:
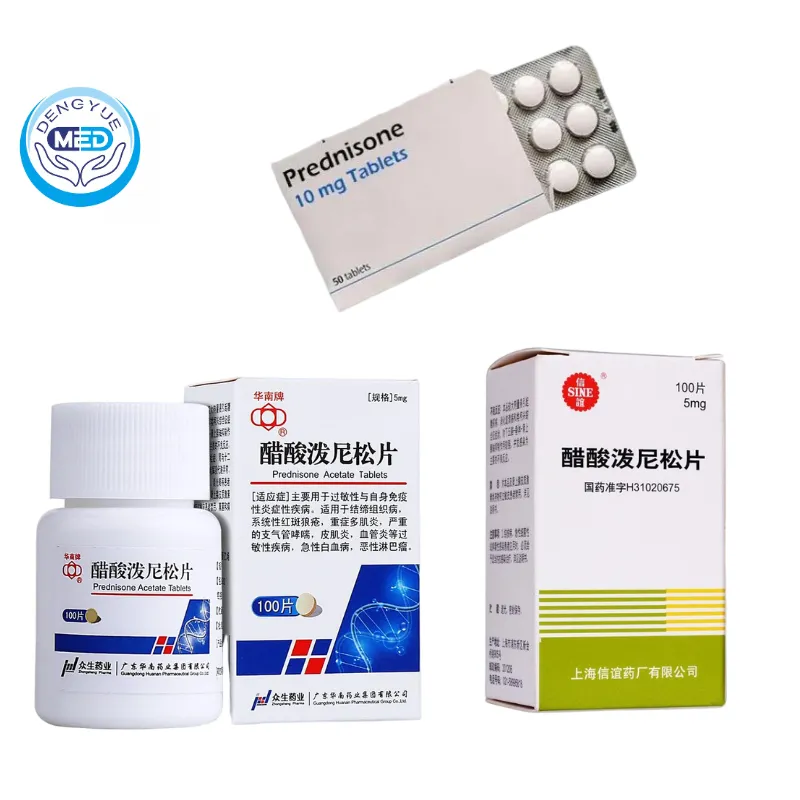
Prednisone
Prednisone is the oldest and most well-documented drug used in the treatment of DMD worldwide. It is one of the core drugs in the standard treatment of DMD and can significantly slow down the decline in motor function and prolong the time it takes to walk independently.
Long-term use is more likely to cause metabolic and skeletal-related adverse reactions such as weight gain, osteoporosis, and growth restriction, requiring standardized monitoring and management.
Deflazacort
Deflazacort has comparable overall efficacy to prednisone, but its effects on weight gain and height, growth, and bone metabolism are relatively mild, making it an important alternative for long-term maintenance therapy.
Long-term side effects such as cataracts or decreased bone mineral density may still occur, and accessibility and cost need to be considered in some regions.
In 2020, Macau launched Defal, a drug for treating DMD. It’s from the same manufacturer as Emflaza in the US, and is the same drug, but due to different countries of market, the price differs by nearly 30 times.

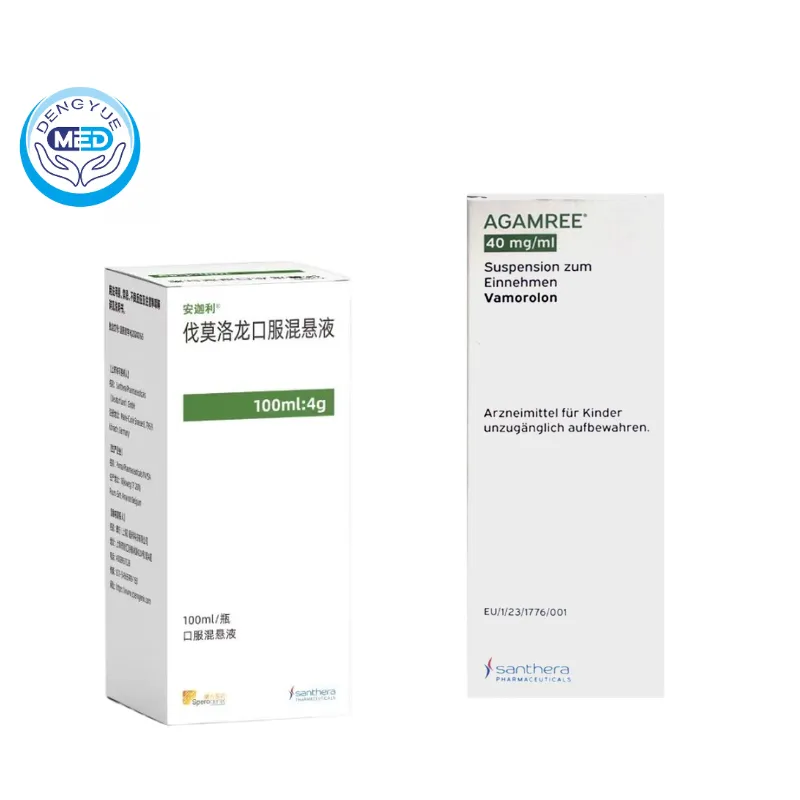
Vamorolone
Vamorolone is a new generation of “uncoupling” steroids that retains anti-inflammatory and muscle-protective effects while minimizing the growth inhibition and bone metabolism effects of traditional glucocorticoids.
Clinical studies show its superior safety profile, making it a more suitable new hormone therapy option for long-term management in children.
✨ These treatment regimens have been shown to extend patients’ independent walking ability by an average of 2-3 years, slow the progression of scoliosis, and postpone the use of ventilators and treatment for heart failure.
However, they do not address the underlying cause, and long-term use can lead to side effects such as weight gain, depression, and osteoporosis.
Exon Skipping Therapy for DMD
Exon skipping therapy represents a significant breakthrough in the drug treatment of Duchenne muscular dystrophy.
Its basic strategy is not to directly repair mutated genes, but rather to regulate the mRNA splicing process, enabling cells to regenerate partially functional dystrophin.
In 2016, the first exon-skipping drug, Eteplirsen (Exondys 51), was approved in the United States for patients with exon 51 skipping mutations, accounting for approximately 10%–15% of all DMD cases, marking the formal entry of the disease into the gene-targeted therapy stage.
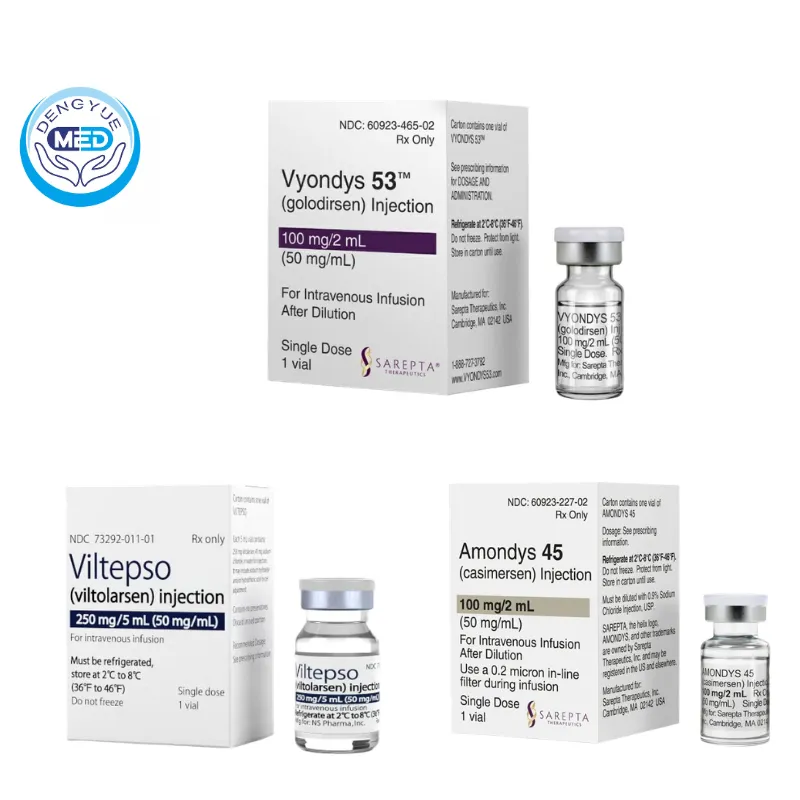
Subsequently, several antisense oligonucleotide therapies targeting different exons have been launched, including:
✅ Golodirsen (Vyondys 53): Targeting patients with exon 53 mutations, further expanding the range of patients eligible for treatment.
✅ Viltolarsen (Viltepso): Also targeting exon 53, clinical studies have shown that it can increase dystrophin expression and improve some motor parameters.
✅ Casimersen (Amondys 45): Targeting exon 45 mutations, providing a new molecular treatment option for another common gene deletion patient group.
Overall, existing exon-skipping drugs collectively cover approximately 30% of DMD patients.
However, many mutation types still lack corresponding targeted therapies, making the development of next-generation molecular therapies with broader site coverage and higher efficacy a key focus of future research.
Gene Therapy for DMD
To expand treatment coverage and intervene in disease progression at the etiological level, gene therapy is becoming a core direction in Duchenne muscular dystrophy research, with several key drugs achieving breakthroughs.
ELEVIDYS (delandistrogene moxeparvovec-rokl)
Received accelerated approval in the US in 2023, and in June 2024, its indication was expanded to all DMD patients ≥4 years of age who are able to walk, with the anti-AAVrh74 antibody restriction removed.
This therapy delivers a miniature dystrophin gene via an AAV vector, aiming to achieve long-term protein expression with a single dose. It is the world’s first approved gene replacement therapy product for DMD.
GNT0004
GNT0004 is currently in Phase III clinical trials, utilizing AAV to deliver a functionally optimized simplified dystrophin gene (hMD1). Its design goal is to improve expression efficiency and stability while reducing the dosage, thereby further improving overall safety.
SGT-003
SGT-003 is still in its early stages, employing a modified miniature dystrophin construct combined with the AAV-SLB101 vector. The research focuses on improving gene delivery efficiency and treatment tolerability.
🐱🏍 Overall, gene replacement therapies, represented by ELEVIDYS, have achieved clinical breakthroughs, while investigational products such as GNT0004 and SGT-003 are driving DMD treatment towards higher safety, better expression efficacy, and broader population coverage.
China’s Drug Development Progress in Duchenne Muscular Dystrophy (DMD)
In China, gene therapies are showing strong momentum in the drug treatment of Duchenne muscular dystrophy, mainly focusing on gene replacement and base editing strategies.
Gene Replacement Therapy
1️⃣ BBM-D101 (Belief BioMed)
Approved for clinical trials by the National Medical Products Administration in April 2025, it is a single intravenous infusion therapy based on engineered AAV vector delivery of optimized dystrophin gene, aiming for long-term expression and sustained efficacy.
Currently in Phase I/II open-label study, primarily evaluating safety, efficacy, and immune response over 52 weeks, with follow-up for up to 5 years of long-term efficacy.
2️⃣ JWK007 (Genevector)
Utilizing a “muscle-targeting capsid + muscle-specific promoter + mini-Dystrophin” design, it aims to improve expression efficiency and safety.
IIT study follow-up showed improved motor function and good tolerability in patients, but it has not yet entered the formal registration clinical stage.
3️⃣ Smart-Dystrophin Gene Therapy (GeneCradle/Byongen Therapeutics)
The two parties entered into a strategic collaboration in 2025 to advance the development of next-generation DMD gene therapy.
This approach aims to improve transgene expression efficiency and overall safety by optimizing the Dystrophin structure and combining it with a novel muscle-targeting AAV capsid. It is currently still in the research and development stage.
4️⃣ Engineered Exosome FL-dystrophin mRNA Therapy (Multi-institutional Collaboration)
Approved in 2025 to initiate a first-in-human clinical trial based on extracellular vesicle delivery of full-length dystrophin mRNA.
This non-viral delivery strategy is expected to provide a new gene therapy pathway in terms of safety and delivery efficiency. It is currently in the investigator-initiated clinical stage.
Base Editing Therapy
1️⃣ GEN6050X (GenAssist)
One of the world’s first DMD base editing drugs to enter clinical trials, suitable for patients who can benefit from exon 50 skipping.
Early clinical data show improvements in exercise and cardiac function, and restoration of dystrophin has been observed. No serious adverse events have been reported so far.
2️⃣ HG302 (HuidaGene Therapeutics)
Based on the high-fidelity CRISPR nuclease hfCas12Max, it achieves precise exon 51 editing and protein restoration through single AAV delivery.
Preliminary clinical results show good safety and early improvement in motor function. A first-in-human multi-dose escalation study is underway.
Overall, innovative DMD treatments in China are developing from AAV gene replacement to a multi-pathway approach combining non-viral delivery and gene editing, showing a continuous trend towards higher safety, more durable efficacy, and broader patient coverage.
Conclusion: Drug Treatment of Duchenne Muscular Dystrophy (DMD) Enters a New Era of Disease-Modifying Therapies
Over the past 40 years, the drug treatment of Duchenne muscular dystrophy has evolved from glucocorticoid-based symptom control to exon skipping and gene therapies with disease-modifying potential, bringing management closer to altering the natural history.
🎇 At the same time, how to ensure that innovative therapies truly benefit patients is becoming a new key challenge.
The service system built around drug accessibility, cross-border supply, and professional medical support is becoming increasingly important. DengYueMed, a Chinese pharmaceutical importer and wholesaler, is driving the faster clinical application of cutting-edge treatments.
Looking to the future, with improved precision in gene editing, the maturity of non-viral delivery, and gradual cost reduction, DMD is expected to transform from a fatal genetic disease into a chronic disease that can be controlled long-term and even has the potential for functional cure.
This medical exploration spanning over a century continues to move forward.
FAQ about Drug Treatment of Duchenne Muscular Dystrophy
What drugs are used to treat Duchenne muscular dystrophy?
Drug treatment of Duchenne muscular dystrophy (DMD) mainly includes corticosteroids (such as prednisone and deflazacort) to slow muscle weakness, as well as mutation-specific therapies like exon-skipping drugs and gene therapy.
What is the latest treatment for DMD?
The latest treatment for Duchenne muscular dystrophy (DMD) includes gene therapy, such as delandistrogene moxeparvovec, which helps restore dystrophin production. New mutation-specific therapies are also emerging.
What is the first line of treatment for muscular dystrophy?
For Duchenne muscular dystrophy (DMD), the first-line treatment is corticosteroids such as prednisone or deflazacort, which help slow muscle degeneration and prolong mobility. Supportive care for heart and respiratory function is also essential.
What is the injection for DMD muscular dystrophy?
Injectable treatments for Duchenne muscular dystrophy (DMD) include exon-skipping drugs given by IV infusion and gene therapy such as delandistrogene moxeparvovec. The option depends on the patient’s specific genetic mutation.



