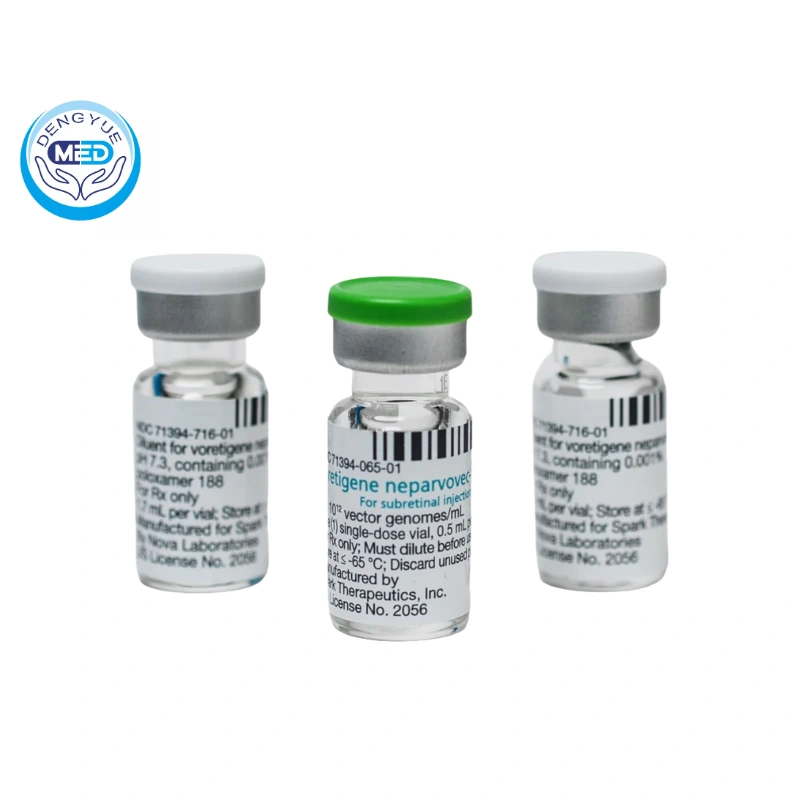
Voretigene Neparvovec-rzyl|Retinitis Pigmentosa|HongKong DengYue Medicine
- Generic Name/Brand Name: Voretigene Neparvovec-rzyl
- Indications: Retinitis Pigmentosa
- Dosage Form: solutions
- Specification: 0.3 mL
Voretigene Neparvovec-rzyl Application Scope
Voretigene neparvovec-rzyl is a gene therapy used to treat retinal dystrophy caused by biallelic RPE65 mutations, including:
- Leber Congenital Amaurosis (LCA) type 2 (66% of cases)
- Early-onset severe retinal dystrophy (EOSRD)
- Retinitis pigmentosa (RP) phenotype (16% of cases).
It is administered via subretinal injection to deliver a functional RPE65 gene to retinal cells, restoring the visual cycle in patients with confirmed mutations.
Voretigene Neparvovec-rzyl Characteristics
- Ingredients: Recombinant adeno-associated virus serotype 2 (AAV2) vector containing human RPE65 cDNA (AAV2-hRPE65v2).
- Properties:
- Single-dose gene therapy.
- Targets retinal pigment epithelium (RPE) cells to restore chromophore production.
- Specification:
- Dose: 1.5 × 10¹¹ vector genomes (vg) in 0.3 mL subretinal injection per eye.
- Packaging Specification: Not detailed in search results.
- Storage: Likely requires cold chain (exact conditions not specified).
- Expiry Date: Not specified.
- Executive Standard: FDA-approved (2017), EMA-approved (2019).
- Approval Number: Not provided.
- Date of Revision: Last FDA label update: May 2024.
- Manufacturer: Novartis (brand name: Luxturna).
Guidelines For The Use Of Voretigene Neparvovec-rzyl
Dosage and Administration:
- Administered as a single subretinal injection per eye, with procedures separated by at least 6 days.
- Requires pars plana vitrectomy for delivery.
- Pre-treatment: Oral steroids (e.g., prednisone) may be given to reduce inflammation.
- Post-treatment: Patients should avoid air travel, high elevations, or scuba diving for ≥1 week due to risk of intraocular air bubbles.
Adverse Reactions:
- Common:
- Conjunctival hyperemia.
- Cataracts, increased intraocular pressure (IOP).
- Retinal tears, macular holes, subretinal deposits.
- Serious:
- Endophthalmitis (eye infection).
- Vision changes (floaters, light flashes, blindness).
Contraindications:
- Hypersensitivity to voretigene or its components.
- Active ocular infections.
Precautions:
- Monitor for retinal detachment, inflammation, or IOP spikes.
- Avoid rubbing eyes; viral shedding in tears may persist for 7 days post-treatment.
Voretigene Neparvovec-rzyl Interactions
Drug Interactions:
- No specific interactions noted, but systemic corticosteroids are often used concurrently to manage inflammation.
- Avoid other retinal therapies that may increase procedural risks (e.g., anticoagulants).
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.
Contact Us




Reviews
There are no reviews yet.