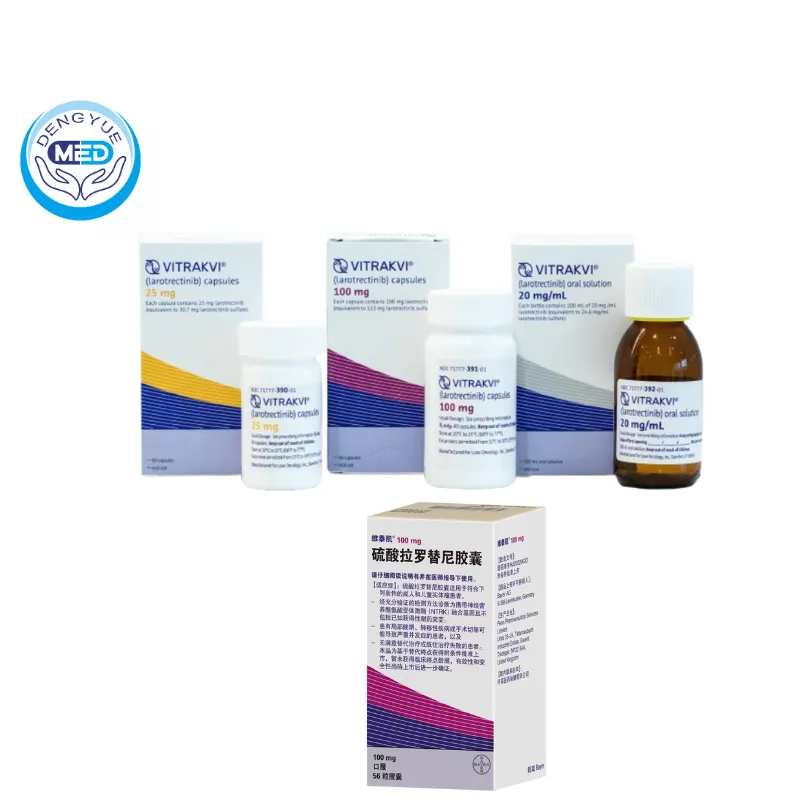
VITRAKVI (Larotrectinib) – Tumor Agnostic Therapy | HongKong DengYue Medicine
- Generic Name/Brand Name: Larotrectinib / VITRAKVI
- Indications: Neurotrophic receptor tyrosine kinase (NTRK) gene fusion
- Dosage Form: Capsules; Oral solution
- Specification: 25 mg/100 mg x 6 Capsules; 20 ml x 1 vial
VITRAKVI Larotrectinib Application Scope
VITRAKVI is indicated for the treatment of adult and pediatric patients with solid tumors that have a neurotrophic receptor tyrosine kinase (NTRK) gene fusion without a known acquired resistance mutation, are either:
Metastatic or where surgical resection is likely to result in severe morbidity, and have no satisfactory alternative treatment options or have progressed following treatment.

VITRAKVI Larotrectinib Characteristics
-
Ingredients: Larotrectinib
-
Properties: A selective tyrosine kinase inhibitor (TKI)
-
Packaging Specification: Available in capsules (e.g., 25 mg and 100 mg strengths, often in bottles of 56 capsules) and as an oral solution (20 mg/mL)
-
Storage:
-
Capsules: Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F). Keep in a dry place, protected from light
-
Oral Solution: Refrigerate at 2°C to 8°C (36°F to 46°F). Do not freeze. Any unused oral solution should be discarded 90 days after first opening the bottle
-
-
Expiry Date: Refer to the expiration date on the packaging.
-
Executive Standard: U.S. NDA: NDA 210861 for larotrectinib (VITRAKVI). Regulatory labeling and updates are available from the FDA
-
Approval Number: U.S. FDA: NDA #211710
-
Date of Revision: See the revision date on the approved product leaflet
-
Manufacturer: Penn Pharmaceutical Services Limited (UK), Orion Corporation (Finland)
Guidelines for the Use of VITRAKVI Larotrectinib
-
Dosage and Administration:
-
Recommended Dose:
-
Adults: 100 mg taken orally twice daily.
-
Pediatric Patients: The recommended dose is 100 mg/m² of body surface area (BSA) taken orally twice daily. The maximum recommended dose per administration is 100 mg
-
-
Administration: Capsules should be swallowed whole with water. Do not chew, crush, or open the capsules due to the bitter taste. The oral solution should be administered using a dedicated oral syringe. It can be taken with or without food. Avoid concomitant administration with grapefruit or grapefruit juice
-
Missed Dose: If a dose is missed, take the next dose at the regular scheduled time. Do not take an extra dose to make up for the missed one. If vomiting occurs after taking a dose, do not take an additional dose; continue with the next scheduled dose
-
-
Adverse Reactions:
-
Common Adverse Reactions: Increased alanine aminotransferase (ALT), increased aspartate aminotransferase (AST), vomiting, constipation, fatigue, nausea, anemia, dizziness, cough, diarrhea, musculoskeletal pain
-
Serious Adverse Reactions: Include neurologic effects (e.g., dizziness, gait disturbance, paresthesia), hepatotoxicity (elevated liver enzymes), and others. Most adverse reactions are Grade 1 or 2 in severity, but Grade 3 or 4 events can occur
-
-
Contraindications: Hypersensitivity to the active substance or any of the excipients
-
Precautions:
-
Confirm NTRK fusion status using validated testing prior to initiating therapy.
-
Monitor liver function tests regularly and interrupt/modify dose for elevated transaminases per label guidance.
-
Use caution in patients with pre-existing hepatic impairment—dose adjustments may be required for moderate to severe hepatic impairment (see clinical pharmacology and dosage modification sections).
-
Be cautious if the patient is taking concomitant medications that are strong CYP3A inducers or inhibitors; larotrectinib is metabolized in part by CYP3A and dose adjustments or monitoring may be required (see Drug Interactions below).
-
VITRAKVI Interactions
-
CYP3A inhibitors (strong): can increase larotrectinib exposure—the product label provides recommended dose reductions when co-administered with strong CYP3A inhibitors.
-
CYP3A inducers (strong): can decrease larotrectinib exposure—co-administration with strong CYP3A inducers should be avoided if possible; dose increases may be recommended if unavoidable (follow label guidance).
-
Consult the full drug interaction tables in the local prescribing information for detailed recommendations and examples of interacting drugs (e.g., ketoconazole, rifampin, certain anticonvulsants, some antiretrovirals, herbal inducers such as St. John’s wort).
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- This content is for reference only. Prescription drugs must be used under a doctor’s guidance and purchased from authorized sources.






Reviews
There are no reviews yet.