Viloxazine|Attention-Deficit/Hyperactivity Disorder(ADHD)
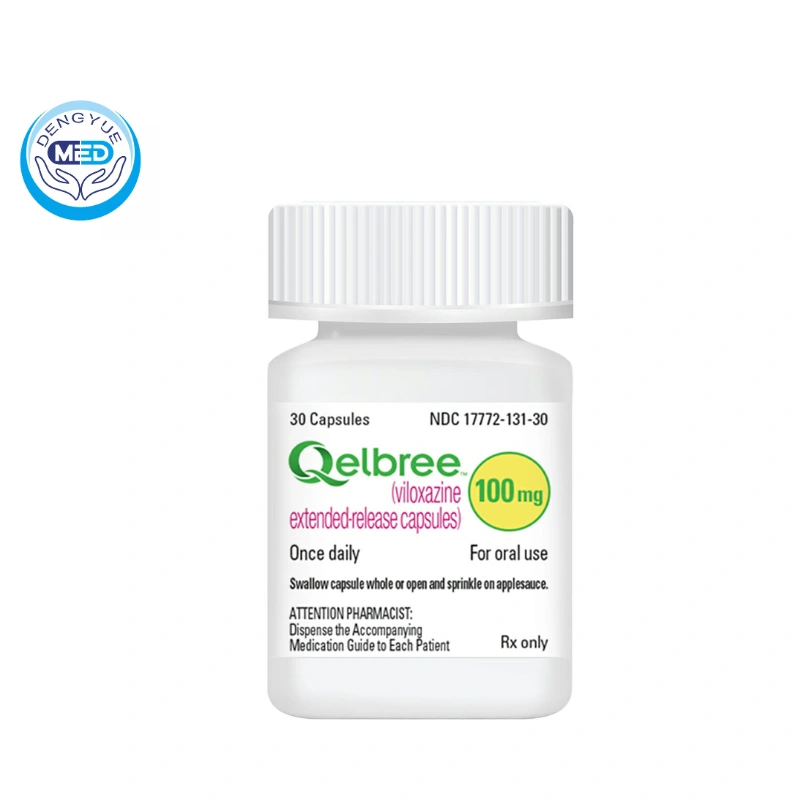
- Generic Name/Brand Name: Viloxazine/Qelbree
- Indications: Attention-Deficit/Hyperactivity Disorder(ADHD)
- Dosage Form: Capsules
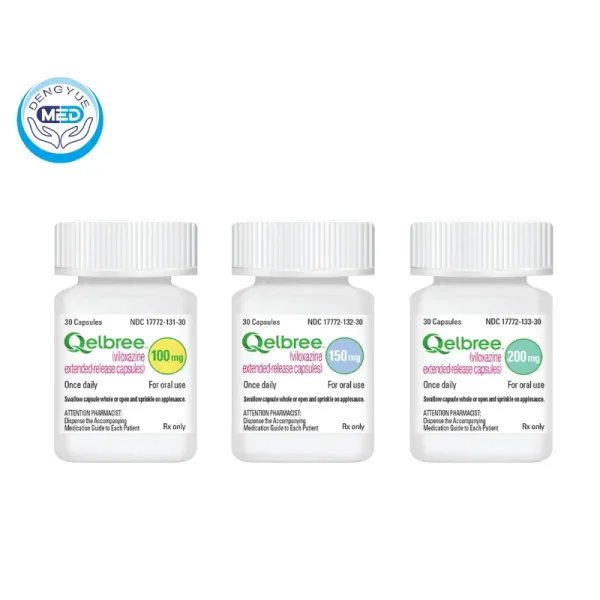
- Specification: 100 mg, 150 mg, and 200 mg strengths
Viloxazine Application Scope
Viloxazine is a selective norepinephrine reuptake inhibitor indicated for the treatment of attention-deficit/hyperactivity disorder(ADHD) in adults and pediatric patients aged 6 years and older.
Viloxazine Characteristics
-
Ingredients:
-
Active Ingredient: Viloxazine hydrochloride.
-
Inactive Ingredients: Sucrose, corn starch, hypromellose, triacetin, polyethylene glycol 3350, lactose monohydrate, titanium dioxide, ethylcellulose, water, ammonia, medium-chain triglycerides, oleic acid, talc, D&C Yellow No. 10, FD&C Yellow No. 6, gelatin, shellac, alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, ferrosoferric oxide, potassium hydroxide.
-
-
Properties:
-
Viloxazine functions as a selective norepinephrine reuptake inhibitor, which is thought to contribute to its therapeutic effects in ADHD.
-
-
Specification:
-
Extended-release capsules available in 100 mg, 150 mg, and 200 mg strengths.
-
-
Packaging Specification:
-
Supplied in bottles containing 30, 60, 90, or 100 capsules.
-
-
Storage:
-
Store at controlled room temperature, 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F).
-
-
Expiry Date:
-
Refer to the packaging for the expiration date.
-
-
Executive Standard:
-
Approved by the U.S. Food and Drug Administration (FDA) under New Drug Application (NDA) number 211964.
-
-
Approval Number:
-
NDA 211964.
-
-
Date of Revision:
-
Refer to the latest prescribing information for the most current revision date.
-
-
Manufacturer:
-
Supernus Pharmaceuticals, Inc.
-
-
Dosage and Administration:
-
Pediatric Patients (6 to 11 years): Start with 100 mg orally once daily; may titrate in increments of 100 mg weekly based on response and tolerability, up to a maximum dose of 400 mg/day.
-
Adolescents (12 to 17 years): Start with 200 mg orally once daily; may titrate in increments of 200 mg weekly based on response and tolerability, up to a maximum dose of 400 mg/day.
-
Adults: Start with 200 mg orally once daily; may titrate in increments of 200 mg weekly based on response and tolerability, up to a maximum dose of 600 mg/day.
-
Capsules may be swallowed whole or opened and sprinkled over applesauce.
-
-
Adverse Reactions:
-
Common adverse reactions include somnolence, fatigue, decreased appetite, and irritability.
-
Monitor for increases in heart rate and blood pressure.
-
Increased risk of suicidal thoughts and behaviors, particularly in pediatric patients; monitor closely for clinical worsening or emergence of suicidal thoughts and behaviors.
-
Contraindications:
-
Concomitant use with monoamine oxidase inhibitors (MAOIs) or within 14 days after discontinuing an MAOI, due to the risk of hypertensive crisis.
-
Concomitant use with sensitive CYP1A2 substrates or CYP1A2 substrates with a narrow therapeutic range.
-
-
Precautions:
-
Suicidal Thoughts and Behaviors: Monitor patients for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of treatment or at times of dose changes.
-
Activation of Mania or Hypomania: Screen patients for bipolar disorder and monitor for signs of mania or hypomania.
-
Cardiovascular Effects: Monitor heart rate and blood pressure regularly; use caution in patients with pre-existing cardiovascular conditions.
-
CNS Depressant Effects: May cause somnolence and fatigue; caution patients against driving or operating hazardous machinery until they know how viloxazine affects them.
-
Viloxazine Interactions
-
Drug Interactions:
-
CYP1A2 Substrates: Viloxazine is a strong inhibitor of CYP1A2; concomitant use with sensitive CYP1A2 substrates or those with a narrow therapeutic range is contraindicated.
-
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.





Reviews
There are no reviews yet.