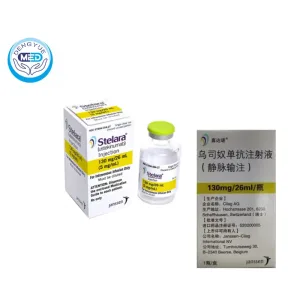
Stelara (Ustekinumab) – Immunomodulator | HongKong DengYue Medicine
- Generic Name/Brand Name: Immunomodulator/Stelara
- Indications: Plaque Psoriasis, Active Psoriatic Arthritis, Crohn’s Disease, Ulcerative Colitis
- Dosage Form: Injection
- Specification: 130 mg/26 mL (5 mg/mL) single×1 vial
Stelara Ustekinumab Application Scope
-
Moderate to severe plaque psoriasis in adults and pediatric patients 6 years of age and older.
-
Active psoriatic arthritis in adults and pediatric patients 6 years of age and older.
-
Moderately to severely active Crohn’s disease (adults).
-
Moderately to severely active ulcerative colitis (adults).
stelara ustekinumab
Stelara Ustekinumab Characteristics
-
Ingredients: Ustekinumab
-
Properties: Sucrose, L-histidine (including L-histidine hydrochloride monohydrate), polysorbate 80, water for injection
-
Packaging Specification: Intravenous infusion (for induction in CD/UC): Vial: 130 mg/26 mL (5 mg/mL)
-
Storage: Refrigerate at 2°C to 8°C. Do not freeze. Keep in original container to protect from light
-
Expiry Date: Pre-filled syringe: 36 months. Vials: 24 months
-
Executive Standard: Complies with relevant biologics manufacturing and quality control standards (e.g., FDA Purple Book listed)
-
Approval Number: US BLA No.: 761044
-
Date of Revision: See the “Revised” or “Last updated” date on the local package insert / prescribing information (product inserts are periodically updated)
-
Manufacturer: Janssen Biotech, Inc. / Janssen-Cilag affiliates (manufacturer names vary by region)
Guidelines for the Use of Stelara Ustekinumab
-
Dosage and Administration:
-
Recommended Dose: PsO (Adults, Subcutaneous):
• Weight ≤100 kg: 45 mg initially, 4 weeks later, then every 12 weeks.
• Weight >100 kg: 90 mg initially, 4 weeks later, then every 12 weeks.
PsO (Pediatric ≥6 years, Subcutaneous): Dose based on body weight.
PsA (Adults, Subcutaneous): 45 mg (or 90 mg for patients >100 kg with inadequate response) at 0, 4 weeks, then every 12 weeks.
CD & UC (Adults, Intravenous Induction → Subcutaneous Maintenance):
• Induction: Single IV infusion dose based on weight (<55 kg: 260 mg; 55-85 kg: 390 mg; >85 kg: 520 mg).
• Maintenance: 90 mg subcutaneously every 8 weeks, starting 8 weeks after IV induction. -
Administration: IV induction must be administered by qualified healthcare professionals in appropriate settings. Subcutaneous injections may be administered by healthcare provider or by trained patient/caregiver after instruction. See full prescribing information for reconstitution and infusion instructions.
-
Missed Dose: If a maintenance dose is missed, administer as soon as possible and then continue with the original dosing schedule as directed by the prescriber. For missed IV induction, contact the treating physician for guidance. (Follow local label recommendations and physician instructions.)
-
-
Adverse Reactions:
-
Common Adverse Reactions: (≥3%): Nasopharyngitis, upper respiratory tract infection, headache, fatigue, injection site reactions (erythema, pain, swelling), nausea, abdominal pain, diarrhea, arthralgia, back pain, pruritus, vomiting
-
Serious Adverse Reactions:
-
Infections: Serious bacterial, fungal, and viral infections; reactivation of latent infections (e.g., tuberculosis). Screen for TB prior to initiation.
-
Malignancies: Observed in clinical trials/postmarketing — monitor as clinically appropriate.
-
Hypersensitivity/anaphylaxis and post-marketing events such as Posterior Reversible Encephalopathy Syndrome (PRES) have been reported.
-
-
-
Contraindications:
-
Known hypersensitivity to ustekinumab or to any excipient in the formulation.
-
Active serious infections (do not start therapy)
-
-
Precautions:
-
Evaluate for active infection before and during treatment. Consider TB screening prior to initiation. Avoid use in patients with active serious infections.
-
Patients receiving STELARA may be at increased risk for infection and malignancy; use clinical judgment when prescribing for patients with chronic or recurrent infections or a history of malignancy.
-
Vaccinations: Do not administer live vaccines during treatment and until immune function has recovered; update immunizations according to guidelines prior to initiation.
-
Stelara Ustekinumab Interactions
- Limited formal drug–drug interaction data. Concomitant use with other immunosuppressive agents may increase risk of infection and should be used with caution. Avoid live vaccines while on therapy. There are no widely reported metabolic enzyme interactions (CYP-mediated) of clinical significance.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- This content is for reference only. Prescription drugs must be used under a doctor’s guidance and purchased from authorized sources.






Reviews
There are no reviews yet.