Ryzneuta (Efbemalenograstim Alfa)- Febrile Neutropenia Prevention
- Generic Name/Brand Name: Efbemalenograstim Alfa/Ryzneuta
- Indications: Chemotherapy-induced febrile neutropenia, Hematology
- Dosage Form: Subcutaneous injection solution
- Specification: 20 mg (1.0 mL) x 1 syringe
Ryzneuta Efbemalenograstim alfa Application Scope
Used to reduce the duration of severe neutropenia and incidence of febrile neutropenia in adults with non-myeloid malignancies undergoing cytotoxic chemotherapy.
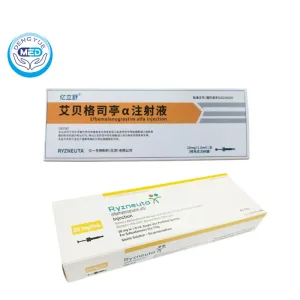
Ryzneuta Efbemalenograstim alfa Characteristics
-
Ingredients:
Efbemalenograstim alfa (recombinant granulocyte colony-stimulating factor fused to human IgG2 Fc)Properties:
Long‑acting G-CSF receptor agonist stimulating bone marrow to produce neutrophils; once‑per‑cycle dosing.Packaging Specification:
20 mg/mL single‑dose prefilled syringe or vialStorage:
2–8 °C; do not freeze or shakeExpiry Date:
Refer to product labelExecutive Standard:
Compliant with local pharmacopeia (e.g., China, US, EU standards)Approval Number:
China NMPA approval in May 2023; FDA approval in Nov 2023; EU in Mar 2024Date of Revision:
[Insert latest label update date]Manufacturer:
Evive Biotechnology (Shanghai) Ltd. (developed), commercialized by regional partners under Ryzneuta brand
Guidelines for the Use of Ryzneuta Efbemalenograstim alfa
-
Dosage and Administration:
-
Recommended Dose: 20 mg SC (single dose) administered once per chemotherapy cycle, ≥24 hours after last chemo, and ≥14 days before next.
-
Administration: Inject subcutaneously; do not administer intravenously.
-
Missed Dose: Administer as soon as possible after missed appointment; continue next cycle per schedule.
-
-
Adverse Reactions:
-
Common Adverse Reactions: Bone pain, back pain, arthralgia, fatigue, headache, fever, nausea, rash, diarrhea
-
Serious Adverse Reactions: Splenic rupture, acute respiratory distress, anaphylaxis, sickle cell crisis, glomerulonephritis, thrombocytopenia, capillary leak syndrome, leukemic transformation in certain high-risk patients
-
-
Contraindications: Known hypersensitivity to G-CSF products or any component of the formulation.
-
Precautions:
-
Monitor for splenic enlargement and pause if severe abdominal pain occurs
-
Use caution in patients with sickle cell disease
-
Monitor pulmonary function; stop treatment if acute respiratory symptoms arise
-
Periodically check blood counts during treatment
-
Ryzneuta Efbemalenograstim alfa Interactions
- No significant drug–drug interactions reported. Avoid use within 24 hours before or after chemotherapy administration.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- This content is for reference only. Prescription drugs must be used under a doctor’s guidance and purchased from authorized sources.




Reviews
There are no reviews yet.