Recombinant Human Somatropin | Growth Hormone Deficiency | DengYueMedicine
- Generic Name: Recombinant Human Somatropin (PEGylated)/Somatrogon
- Indications: Growth hormone deficiency
- Dosage Form: Injection
- Specification:10 mg, 20 mg
Recombinant Human Somatropin Application Scope
Recombinant Human Somatropin is primarily used for the treatment of growth hormone deficiency (GHD) in both children and adults. It is also used in conditions such as:
- Short stature associated with idiopathic growth hormone deficiency or turner syndrome.
- Chronic kidney disease (CKD) in children where growth failure is observed.
- Prader-Willi syndrome (a genetic disorder leading to developmental delays and short stature).
- Growth failure in children due to other causes such as low birth weight or hypopituitarism.
- Adult growth hormone deficiency, particularly in patients with a history of pituitary tumors or radiation therapy affecting the pituitary gland.
Recombinant Human Somatropin helps promote growth in children by stimulating growth plates in the bones and enhancing protein synthesis, while in adults, it may help improve muscle mass, reduce fat mass, and increase bone density.
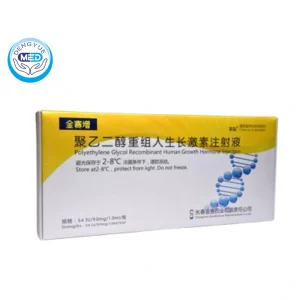
Recombinant Human Somatropin Characteristics:
- Ingredients:The active ingredient is Recombinant Human Growth Hormone (Somatropin). The drug is conjugated with Polyethylene Glycol (PEG) to increase its stability and extend its duration of action.
- Properties:This formulation is a long-acting human growth hormone analog that works by stimulating growth and cellular regeneration. PEGylation reduces the frequency of injections needed by extending the half-life of somatropin.
- Specification:
Typically available in concentrations such as 10 mg, 20 mg, or other dosages based on the specific product formulation. The exact dosage strength and concentration should be listed by the manufacturer.
Packaging Specification:
- Storage:
- Store in a refrigerator at 2°C to 8°C (36°F to 46°F).
- Do not freeze. If the drug has been frozen, it should not be used.
- Keep the product in the original packaging to protect it from light.
- After reconstitution or opening (if applicable), follow manufacturer-specific guidelines for storage duration.
- Effective Period:
- The shelf life of the product is typically 1 to 2 years from the manufacturing date, and it should be used before the expiration date marked on the packaging.
- Approval Number:
- The approval number varies by country (e.g., FDA approval in the U.S., EMA approval in the EU, or others). For specific approval details, consult the product’s official documentation or manufacturer’s information.
- Manufacturer:
- The manufacturer for Polyethylene Glycol Recombinant Human Somatropin Injection may vary depending on the market.
Guidelines For The Use Of Recombinant Human Somatropin
- Dosage and Administration:
- The dosage is typically individualized based on the patient’s age, weight, and specific condition being treated.
- For children with growth hormone deficiency, the initial dose is usually between 0.016 to 0.05 mg/kg/day, which can be adjusted based on response.
- In adults with growth hormone deficiency, the starting dose is generally around 0.2 to 0.3 mg per day, with adjustments as necessary.
- The injection is typically administered subcutaneously once a week or as per the medical advice.
- Adverse Reactions:
- Common side effects may include pain at the injection site, headache, muscle pain, and swelling.
- Rare but serious adverse effects include increased risk of diabetes, fluid retention, and joint issues.
- Long-term use may also increase the risk of certain cancers, particularly in patients with pre-existing risk factors.
Medication Limitations
- Contraindications:
- Hypersensitivity to recombinant human growth hormone or any of the excipients in the formulation.
- Active malignancy or other conditions where growth hormone therapy could stimulate unwanted cell proliferation.
- Prader-Willi syndrome patients with severe obesity or respiratory distress are contraindicated for this therapy.
- Precautions:
- Caution is needed in patients with diabetes or insulin resistance.
- Monitor growth hormone-treated patients regularly for adverse effects, including fluid retention, glucose intolerance, and changes in thyroid function.
- Regular monitoring of height, weight, and bone age in pediatric patients is necessary.
Recombinant Human Somatropin Interactions
- Drug Interactions:
- Corticosteroids may reduce the effectiveness of recombinant human growth hormone therapy.
- Insulin and oral antidiabetic agents may require dosage adjustments in patients receiving growth hormone therapy.
- Care should be taken when co-administering with other growth factors or cytokines, as interactions can lead to excessive growth effects or other adverse events.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from HongKong DengYue Medicine. It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.
Contact Us




Reviews
There are no reviews yet.