Leqvio (Inclisiran) | ASCVD | HongKong DengYue Medicine
- Generic Name/Brand Name: Inclisiran/Leqvio
- Indications: ASCVD or heterozygous familial hypercholesterolemia (HeFH)
- Dosage Form: SC injection solution
- Specification: 284 mg/1.5 mL prefilled syringe (Single-dose syringe; 189 mg/mL)
Inclisiran Application Scope
Inclisiran (Leqvio) is indicated for adult patients with elevated LDL‑C—due to primary hypercholesterolemia, HeFH, ASCVD, or elevated cardiovascular risk—as an adjunct to lifestyle and statin-based therapy, with an indication expanded in July 2023 to include earlier intervention in high-risk individuals without prior events.
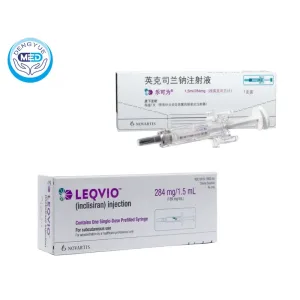
Inclisiran Characteristics
-
Ingredients:
-
Active: Synthetic small-interfering RNA (siRNA) targeting PCSK9 mRNA
-
Inactive: Formulation-specific excipients (proprietary per FDA label)
-
-
Properties:
-
Specifically harnesses RNA interference to reduce hepatic PCSK9 production → increases LDL receptors → ~50 % LDL‑C reduction
-
Restricted to liver, no impact on DNA, half-life ~9 hours but prolonged effect due to RISC activity
-
-
Packaging Specification: Single-dose, prefilled syringe containing 284 mg/1.5 mL (189 mg/mL) clear, colorless to pale yellow solution
-
Storage: Refrigerated at 2–8 °C; may be kept at room temperature (≤ 25 °C) up to 30 days before use
-
Expiry Date: (Shelf life) 36 months (3 years) from manufacture
-
Executive Standard: FDA-approved as a biologic (BLA 125562); meets European Medicines Agency (EMA) and WHO criteria
-
Approval Number: FDA: BLA 125562, approved December 22, 2021
-
Date of Revision: Label updates ongoing; safety and dosing information current as of early 2024
-
Manufacturer: Novartis Pharmaceuticals Corp.
Guidelines for the Use of Inclisiran
-
Dosage and Administration:
-
Initial: 284 mg SC injection on Day 1, then at 3 months, followed by every 6 months
-
Administered by a healthcare professional in the thigh, abdomen, or upper arm
-
No dose adjustments needed for renal or mild/moderate hepatic impairment
-
-
Adverse Reactions:
-
≥ 3% and more frequent than placebo: injection-site reactions (8%), arthralgia (5%), bronchitis (4%), urinary tract infection (4.4%), diarrhea (3.9%), extremity pain (3.3%), dyspnea (3.2%)
-
Hypersensitivity including angioedema (rare)
-
-
Contraindications: History of serious hypersensitivity reaction to Inclisiran or excipients (e.g., injection components)
-
Precautions:
-
Monitor for hypersensitivity reactions post-injection
-
Use caution in severe hepatic or renal impairment (limited data)
-
No routine lab monitoring mandated beyond standard lipid panels and safety labs
-
Inclisiran Interactions
- Drug Interactions:
-
No known clinically meaningful interactions with CYP substrates.
-
Safe to use with statins, ezetimibe, PCSK9 inhibitors (antibodies)
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.



Reviews
There are no reviews yet.