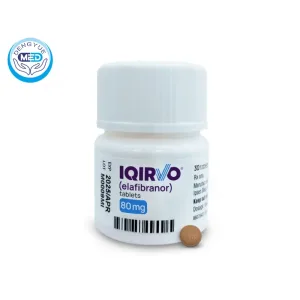
Iqirvo (Elafibranor) | Primary Biliary Cholangitis | DengYue Medicine
- Generic Name/Brand Name: Elafibranor/ Iqirvo
- Indications: primary biliary cholangitis (PBC) in adults, in combination with UDCA or as monotherapy
- Dosage Form: Film-coated tablet
- Specification: 80 mg per tablet
Elafibranor Application Scope
Elafibranor (Iqirvo) is indicated for the treatment of:
-
Primary Biliary Cholangitis (PBC) in adults:
-
In combination with ursodeoxycholic acid (UDCA) in patients with an inadequate response to UDCA.
-
As monotherapy in patients unable to tolerate UDCA.
-
This indication is approved under accelerated approval based on reduction of alkaline phosphatase (ALP). Continued approval may be contingent upon verification of clinical benefit in confirmatory trials.
Characteristics
-
Ingredients:
-
Active Ingredient: Elafibranor
-
Chemical Formula: C₂₂H₂₄O₄S
-
Molecular Weight: 384.14 g/mol
-
Mechanism of Action: Dual peroxisome proliferator-activated receptor (PPAR) α/δ agonist, believed to work by inhibiting bile acid synthesis through activation of PPAR-alpha and PPAR-delta.
-
-
Properties: Elafibranor is a dual peroxisome proliferator-activated receptor (PPAR) α and δ agonist, primarily developed for the treatment of primary biliary cholangitis (PBC).
-
Packaging Specification: Supplied in child-resistant bottles containing 30 tablets.
-
Storage:
-
Store at room temperature between 15°C to 30°C (59°F to 86°F).
-
Keep in the original package to protect from moisture and light.
-
-
Expiry Date: Refer to the packaging for the expiration date.
-
Executive Standard: Approved under FDA regulations.
-
Approval Number: FDA approval granted in June 2024
-
Date of Revision: Refer to the latest prescribing information for revision dates.
-
Manufacturer: Ipsen Biopharmaceuticals, Inc.
Guidelines for the Use of Elafibranor
-
Dosage and Administration:
-
Recommended Dosage: 80 mg taken orally once daily, with or without food.
-
Administration with Bile Acid Sequestrants: Administer Elafibranor at least 4 hours before or 4 hours after taking a bile acid binding sequestrant, or at as great an interval as possible.
-
-
Adverse Reactions:
Common side effects include:
-
Myalgia, myopathy, and rhabdomyolysis.
-
Fractures.
-
Drug-induced liver injury.
-
Hypersensitivity reactions.
-
-
Contraindications: Unknown.
-
Precautions:
-
Muscle-related Issues: Assess for myalgia and myopathy before initiating treatment; monitor during treatment.
-
Fracture Risk: Monitor bone health according to standard care.
-
Embryo-fetal Toxicity: Verify negative pregnancy test before initiating in females of reproductive potential.
-
Liver Injury: Monitor liver function tests; interrupt treatment if liver tests worsen.
-
Hypersensitivity Reactions: Monitor for signs; discontinue if severe reactions occur.
-
Biliary Obstruction: Avoid use in patients with complete biliary obstruction.
-
Interactions
- Drug Interactions:
-
Hormonal Contraceptives: Elafibranor may reduce the effectiveness of hormonal contraceptives. Use non-hormonal methods during treatment and for at least 3 weeks after the last dose.
-
HMG-CoA Reductase Inhibitors (Statins): Increased risk of myopathy; monitor for muscle-related symptoms.
-
Rifampin: May reduce Elafibranor exposure; monitor liver enzymes if co-administered.
-
Bile Acid Sequestrants: May reduce Elafibranor absorption; administer Elafibranor at least 4 hours before or after these agents.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.





Reviews
There are no reviews yet.