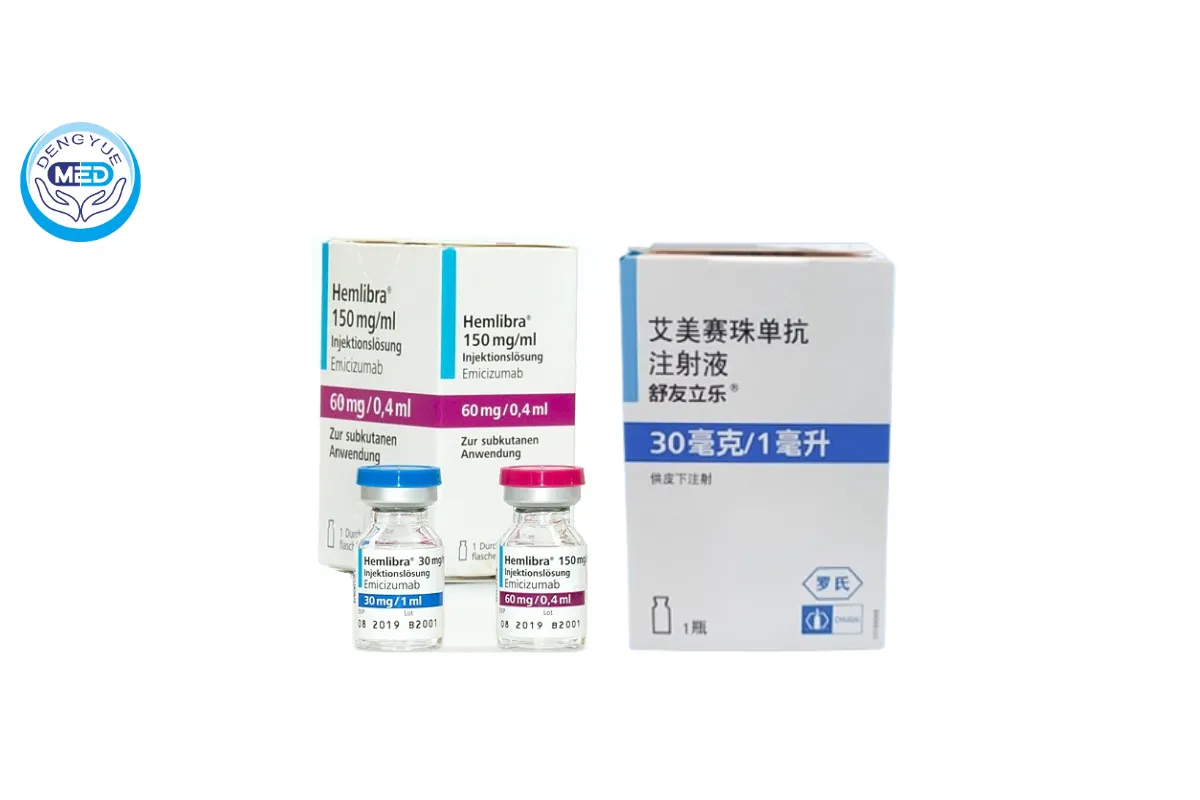
Hemlibra (Emicizumab) – Hemophilia A | HongKong DengYue Medicine
- Generic Name/Brand Name: Hemlibra (Emicizumab)
- Indications: Hemophilia A
- Dosage Form: 3 mg/kg once weekly for the first 4 weeks.
- Specification: 30 mg (1 mL)* 1 vial
Emicizumab Application Scope
Emicizumab is indicated for routine prophylaxis to prevent or reduce the frequency of bleeding episodes in adults and children with congenital factor VIII deficiency (Hemophilia A) who:
Have factor VIII inhibitors, or
Have severe hemophilia A (FVIII activity < 1%) without inhibitors.
Emicizumab Characteristics
-
Ingredients:
Emicizumab (recombinant, produced in Chinese hamster ovary [CHO] cells)
- Properties:
A colorless to slightly yellow solution, sterile and preservative-free.
-
Packaging Specification:
30 mg (1 mL)/vial
60 mg (0.4 mL)/vial
105 mg (0.7 mL)/vial
150 mg (1 mL)/vial
-
Storage:
Store in a refrigerator at 2 °C–8 °C.
Do not freeze. Protect from light. Keep the vial in the original carton until use.
-
Expiry Date:
36 months from the date of manufacture (refer to the packaging label for the exact date
-
Executive Standard:
Manufactured in accordance with China NMPA therapeutic biological product standards for monoclonal antibody preparations.
-
Approval Number:
NMPA Approval No.: S20180025 / S20180026 / S20180027
-
Date of Revision:
August 24, 2023
- Manufacturer:
Shanghai Roche Pharmaceuticals Ltd.
Authorized by Roche Pharma (Schweiz) AG, Switzerland
Guidelines for the Use of xxx
-
Dosage and Administration:
-
Recommended Dose: 3 mg/kg once weekly for the first 4 weeks.
-
Administration: Administer under the supervision of a healthcare professional experienced in the treatment of hemophilia.
-
Missed Dose: If a dose is missed, it should be administered as soon as possible and the usual dosing schedule resumed.
-
-
Adverse Reactions:
-
Common Adverse Reactions:
-
Injection site reactions (redness, itching, discomfort)
Headache
Arthralgia
Fever
Fatigue
-
-
Serious Adverse Reactions:
-
Thrombotic microangiopathy (TMA)
-
Thromboembolic events, particularly when used concurrently with activated prothrombin complex concentrate (aPCC) or PCC at high cumulative doses
-
-
-
Contraindications:
Contraindicated in patients with a known hypersensitivity to Emicizumab or any of its excipients.
-
Precautions:
-
Use caution when combining Emicizumab with bypassing agents (especially aPCC or PCC) due to increased risk of thrombotic microangiopathy or thrombosis.
-
Emicizumab may interfere with certain coagulation assays (such as APTT), leading to inaccurate results; specialized testing methods should be used.
-
Should be administered only under the guidance of physicians experienced in managing hemophilia and bleeding disorders.
-
Emicizumab Interactions
- Concomitant use with activated prothrombin complex concentrate (aPCC) or prothrombin complex concentrate (PCC) can increase the risk of thrombosis or TMA.
-
No clinically significant pharmacokinetic drug–drug interactions have been reported with other agents.
- Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- This content is for reference only. Prescription drugs must be used under a doctor’s guidance and purchased from authorized sources.




.webp)

Reviews
There are no reviews yet.