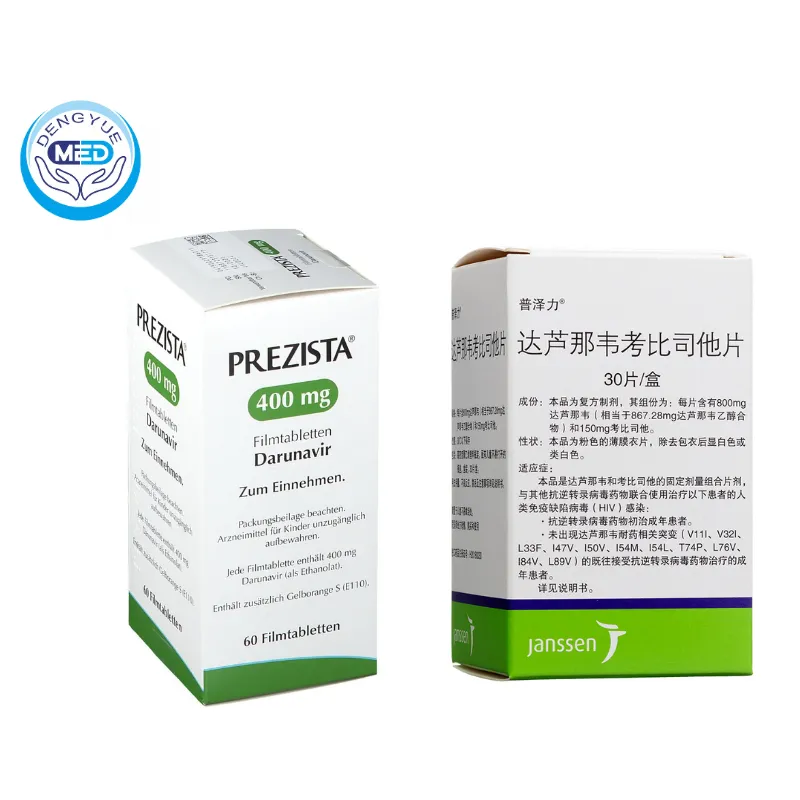
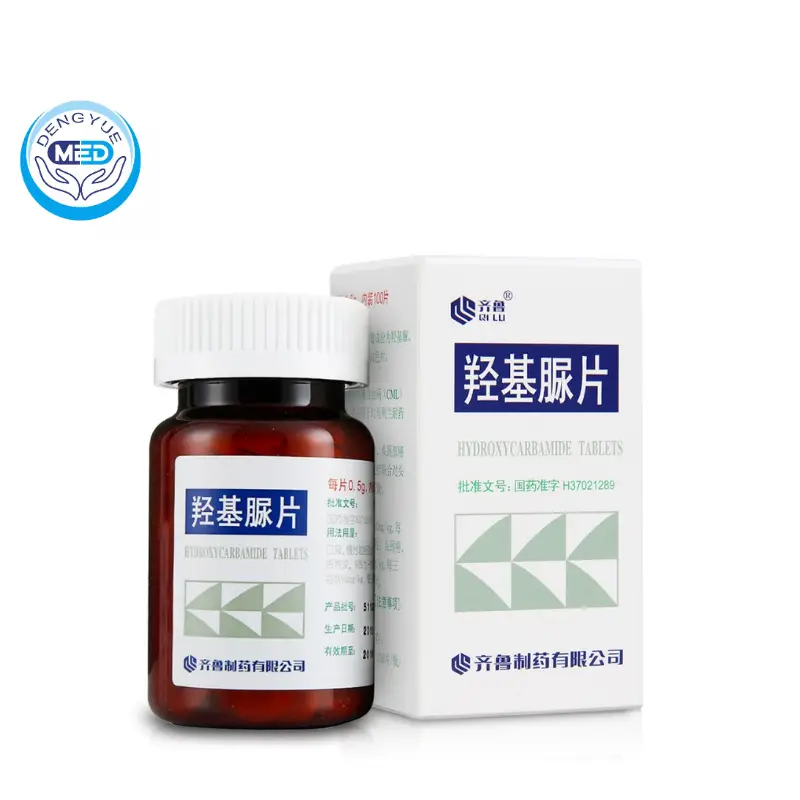
Darunavir/Cobicistat|HIV-1 infection|HongKong DengYue
- Generic Name/Brand Name:Darunavir/Rezolsta
- Indications: HIV-1 infection
- Dosage Form: Film-coated tablet
- Specification: 800 mg/150 mg
Darunavir/Cobicistat Application Scope
Is a fixed-dose combination antiretroviral medication used in the treatment of HIV-1 infection in adults and pediatric patients (typically aged ≥12 years and weighing ≥40 kg).
- Darunavir: A protease inhibitor (PI) that blocks HIV viral replication.
- Cobicistat: A pharmacokinetic enhancer (CYP3A4 inhibitor) that boosts darunavir levels.
Characteristics
Ingredients:
- Active: Darunavir ethanolate (equivalent to darunavir 800 mg) + cobicistat 150 mg per tablet.
- Inactive: May include microcrystalline cellulose, silicon dioxide, etc.
Properties:
- Appearance: Film-coated tablet (e.g., yellow, oval).
- Solubility: Poorly soluble in water.
Specification:
- Available as 800 mg darunavir/150 mg cobicistat per tablet.
Packaging Specification:
- Bottles of 30 tablets or blister packs.
Storage:
- Store below 30°C (86°F), protect from moisture.
Expiry Date:
- Typically 24 months from manufacture (check packaging).
Executive Standard:
- Complies with USP (United States Pharmacopeia) or EMA (European Medicines Agency) standards.
Approval Number:
- FDA: e.g., NDA 205395 (Prezista/cobicistat).
- EMA: EU/1/06/347/001 (check local regulations).
Date of Revision:
- Varies by manufacturer (e.g., latest label updates in 2023).
Manufacturer:
- Janssen Pharmaceuticals (Johnson & Johnson).
Guidelines for the Use
Dosage and Administration:
- Adults: 1 tablet (800/150 mg) once daily with food.
- Pediatrics: Weight-based dosing (≥40 kg).
- Must be combined with other antiretrovirals .
Adverse Reactions:
- Common: Diarrhea, nausea, rash, headache.
- Serious: Severe skin reactions (e.g., Stevens-Johnson syndrome), hepatotoxicity, hyperglycemia.
Contraindications:
- Hypersensitivity to darunavir/cobicistat.
- Concurrent use with CYP3A substrates .
Precautions:
- Monitor liver function (risk of hepatitis B/C flare-up).
- May increase serum creatinine (cobicistat effect).
- Pregnancy: Use only if benefits outweigh risks (Category C).
Interactions
Drug Interactions:
- Contraindicated:
- CYP3A inducers: Rifampin, carbamazepine.
- Alpha-1-adrenergic antagonists: Alfuzosin (severe hypotension).
- Herbal supplements: St. John’s wort.
- Require Dose Adjustment:
- Statins: Avoid simvastatin/lovastatin; use atorvastatin with caution.
- Anticoagulants: Monitor warfarin closely.
- Anticonvulsants: Phenytoin, phenobarbital.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.
Contact Us


Reviews
There are no reviews yet.