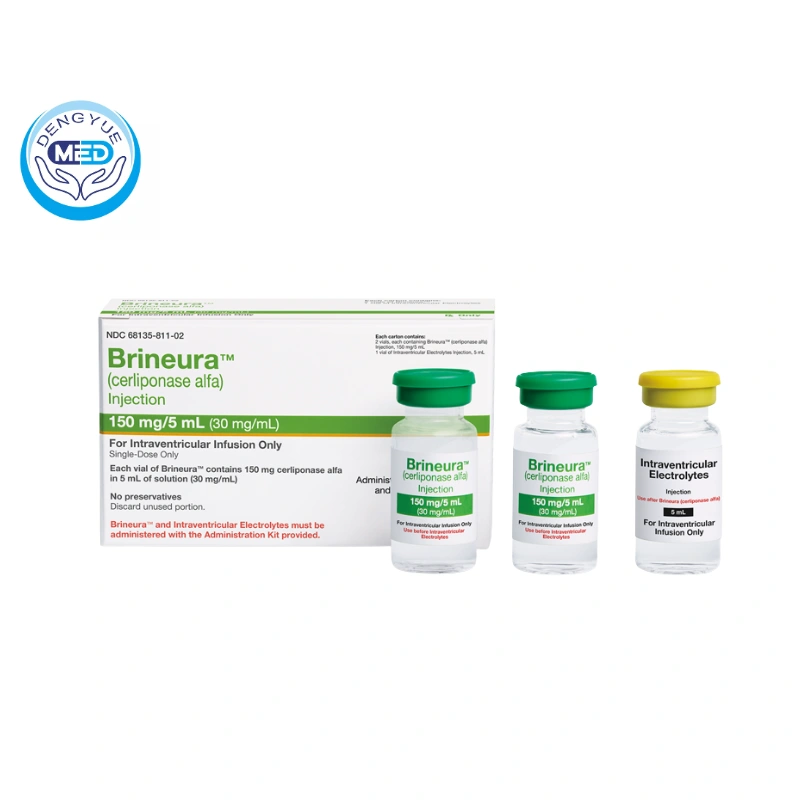
Cerliponase Alfa|CLN2 Disease|HongKong DengYue Medicine
- Generic Name/Brand Name: Cerliponase alfa/Brineura
- Indications: Symptomatic CLN2 disease (≥3 years old)
- Dosage Form: Intraventricular infusion solution
- Specification: 150 mg/5 mL per vial
Cerliponase Alfa Application Scope
Cerliponase alfa is an enzyme replacement therapy used for:
- Late-infantile neuronal ceroid lipofuscinosis type 2 (CLN2 disease) – A rare, fatal neurodegenerative lysosomal storage disorder caused by tripeptidyl peptidase-1 (TPP1) deficiency.
- Symptomatic children ≥3 years old – Slows the decline of motor function, particularly walking ability.
Characteristics
Ingredients
- Active: Recombinant human TPP1 enzyme (cerliponase alfa).
- Excipients: Stabilizers in the intraventricular infusion solution (exact formulation not publicly detailed).
Properties
- Mechanism: Binds to mannose-6-phosphate receptors (CI-MPR) in the CNS, delivering TPP1 to lysosomes to degrade lipofuscin deposits.
- Molecular Weight: ~65 kDa (glycosylated form).
Specification
- 30 mg/mL (intraventricular infusion solution).
Packaging Specification
- Single-dose vial (150 mg/5 mL).
Storage
- Unopened vials: 2–8°C (refrigerated) – Do not freeze.
- After reconstitution: Stable for ≤24 hours if refrigerated.
Expiry Date
- Typically 24 months when stored properly (check batch-specific labeling).
Executive Standard
- Manufactured under cGMP (FDA/EMA-approved).
Approval Number
- FDA: Approved April 2017 (first treatment for CLN2).
- EMA: Approved 2017 (EU authorization).
Date of Revision
- Varies by region (latest US label update: 2023).
Manufacturer
- BioMarin Pharmaceutical Inc. (brand name: Brineura®).
Guidelines for Use Of Cerliponase Alfa
Dosage and Administration
- Recommended dose: 300 mg every other week via intraventricular infusion (via implanted reservoir).
- Infusion protocol:
- Premedicate with antihistamines/antipyretics ± corticosteroids (30–60 min before).
- Administer over ~4.5 hours, followed by intraventricular electrolyte infusion.
- Hospital administration required (due to infection/seizure risks).
Adverse Reactions
- Common (≥10%): Fever, vomiting, seizures, CSF protein increase, hypersensitivity, headache.
- Serious:
- Device-related infections.
- Cardiovascular events (bradycardia, hypotension).
Contraindications
- Active CNS/device-site infections.
- Acute intraventricular device complications.
Precautions
- Infusion monitoring: ECG for cardiac abnormalities; CSF sampling pre-infusion.
- Hypersensitivity: Immediate discontinuation if anaphylaxis occurs.
- Seizure risk: Monitor neurological status closely.
Interactions
Drug Interactions
- No major interactions reported, but concomitant CNS-depressants may exacerbate seizures.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.
Contact Us




Reviews
There are no reviews yet.