Beyfortus (Nirsevimab) – Respiratory Syncytial Virus | HongKong DengYue Medicine
- Generic Name/Brand Name: Nirsevimab/Beyfortus
- Indications: RSV
- Dosage Form: Syringe
- Specification: 50 mg & 100 mg × 1
Nirsevimab Application Scope
Prevention of respiratory syncytial virus (RSV) lower respiratory tract disease in: Neonates and infants born during or entering their first RSV season. Children up to 24 months of age who remain vulnerable to severe RSV disease through their second RSV season.
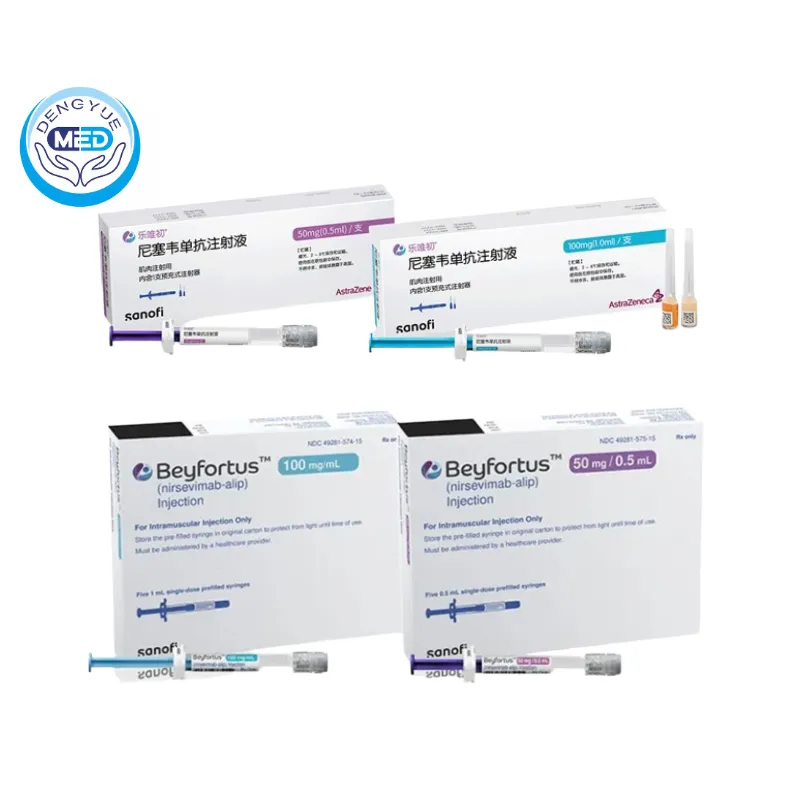
Nirsevimab Characteristics
-
Ingredients: Active ingredient: nirsevimab-alip. Inactive ingredients: arginine hydrochloride, histidine, L-histidine hydrochloride monohydrate, polysorbate 80, sucrose and water for injection.
-
Properties: BEYFORTUS (nirsevimab-alip) injection is a sterile, preservative-free, clear to opalescent, colorless to yellow solution for intramuscular injection. Nirsevimab-alip, a respiratory syncytial virus F protein-directed fusion inhibitor, is a human immunoglobulin G1 kappa (IgG1 κ ) monoclonal antibody produced in Chinese hamster ovary (CHO) cells by recombinant DNA technology. The molecular weight is approximately 146.3 kDa.
-
Packaging Specification: BEYFORTUS injection is supplied as follows: • One 50 mg/0.5 mL single-dose pre-filled syringe in a carton: NDC 49281-575-00 • Five 50 mg/0.5 mL single-dose pre-filled syringes in a carton: NDC 49281-575-15 • One 100 mg/mL single-dose pre-filled syringe in a carton: NDC 49281-574-88 • Five 100 mg/mL single-dose pre-filled syringes in a carton: NDC 49281-574-15 Each BEYFORTUS pre-filled syringe is for one time use only.
-
Storage: Store refrigerated between 36°F to 46°F (2°C to 8°C). BEYFORTUS may be kept at room temperature 68°F to 77°F (20°C to 25°C) for a maximum of 8 hours. After removal from the refrigerator, BEYFORTUS must be used within 8 hours or discarded. Store BEYFORTUS in original carton to protect from light until time of use. Do not freeze. Do not shake. Do not expose to heat.
-
Expiry Date: Do not use if the BEYFORTUS pre-filled syringe has been dropped or damaged, the security seal on the carton has been broken, or the expiration date has passed.
-
Executive Standard: J06BD08 (Antiviral monoclonal antibodies)
-
Approval Number: Initial U.S. Approval: 761328
-
Date of Revision: 08/2024
-
Manufacturer: AstraZeneca and Sanofi.
Guidelines for the Use of Nirsevimab
-
Dosage and Administration:
-
Recommended Dose: Neonates and infants born during or entering their first RSV season: • 50 mg if less than 5 kg in body weight. • 100 mg if greater than or equal to 5 kg in body weight. Children who remain vulnerable through their second RSV season: • 200 mg (2 x 100 mg injections). For children undergoing cardiac surgery with cardiopulmonary bypass, an additional dose may be required based on timing since previous dose.
-
Administration: Give as intramuscular injection by a healthcare provider, preferably in thigh front. Avoid gluteal muscle. Check for clarity before use; discard if cloudy or particles present. Use separate sites for multiple injections. Can be given with childhood vaccines at different sites; do not mix in same syringe. Avoid palivizumab in same season if already given Nirsevimab.
-
Missed Dose: N/A
-
-
Adverse Reactions:
-
Common Adverse Reactions: Most common adverse reactions were rash (0.9%) and injection site reactions (0.3%).
-
Serious Adverse Reactions: Serious hypersensitivity reactions have been reported following BEYFORTUS administration. These reactions included urticaria, dyspnea, cyanosis, and/or hypotonia. Anaphylaxis has been observed with human immunoglobulin G1 (IgG1) monoclonal antibodies.
-
-
Contraindications: BEYFORTUS is contraindicated in infants and children with a history of serious hypersensitivity reactions, including anaphylaxis, to nirsevimab-alip or to any of the excipients.
-
Precautions:
- As with any other IM injections, BEYFORTUS should be given with caution to infants and children with thrombocytopenia, any coagulation disorder, or to individuals on anticoagulation therapy. The safety and effectiveness of BEYFORTUS in children older than 24 months of age have not been established.
Nirsevimab Interactions
- No interference with RT-PCR or rapid RSV tests targeting sites I, II, or IV. Use RT-PCR to confirm if symptoms suggest RSV but test negative. No formal drug studies; unlikely to affect enzymes or transporters. Limited data on co-use with vaccines shows similar safety; no data with other immunoglobulins.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- This content is for reference only. Prescription drugs must be used under a doctor’s guidance and purchased from authorized sources.

.webp)



Reviews
There are no reviews yet.