
FDA Accelerates Approval Of TALVEY™ (Talquetamab-tgvs) For The Treatment Of Multiple Myeloma
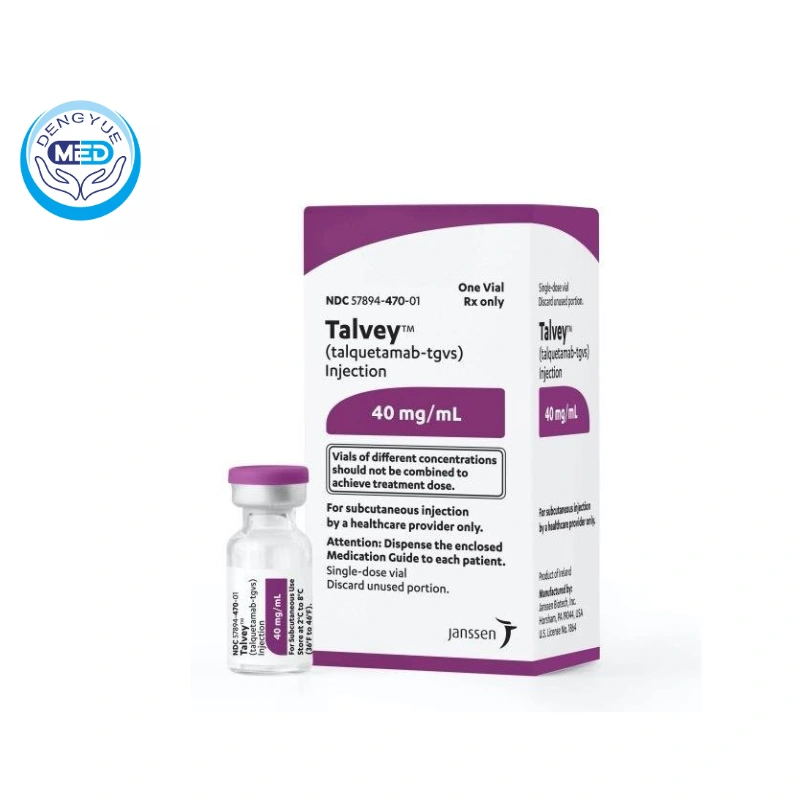
On 9 August 2023, the US Food and Drug Administration (FDA) announced the accelerated approval of TALVEY™ (generic: talquetamab-tgvs) for the treatment of adult patients with relapsed or refractory multiple myeloma (RRMM).
These patients will need to have received at least four prior therapies, including proteasome inhibitors, immunomodulators and anti-CD38 monoclonal antibodies.
TALVEY Clinical Trial
Talvey’s clinical trial results showed significant efficacy in the treatment of multiple myeloma. In a clinical trial of patients with multiple myeloma, patients in Talvey’s treatment group had significantly better overall survival and progression-free survival than the control group.
In addition, the safety and tolerability of Talvey has been well established, providing patients with a safer and more effective treatment option.
Study Findings
Talvey’s research and development team said they will continue to conduct in-depth studies on the drug’s mechanism of action and clinical applications, with a view to providing better treatment options for multiple myeloma patients.
They also hope to collaborate with medical institutions and research teams around the world to advance the field of multiple myeloma treatment.
Safety Tips
Common adverse reactions include cytokine release syndrome (CRS), oral-related symptoms (e.g., dysgeusia, dry mouth), and infections, and the FDA recommends step-dose escalation prior to use to reduce the risk of CRS.
Looking Ahead
For multiple myeloma patients, the advent of Talvey talquetamab-tgvs is undoubtedly a boon. It not only provides patients with a new treatment option, but also offers them new hope.
Talvey will complement Johnson & Johnson/Legend Bio’s Carvykti (BCMA CAR-T), further strengthening the company’s leadership position in multiple myeloma.
It is believed that in the future, with the emergence of Talvey and other innovative drugs, the treatment of multiple myeloma will make even more significant progress.
However, DengYue’s efforts to increase the affordability of the medicine are critical to ensuring that more patients benefit from it.
We welcome you to reach out to us to buy Talquetamab-tgvs. DengYueMedicine is here to provide detailed medicine information, pricing, and support to ensure a smooth and reliable buying experience.
Feel free to contact us anytime to discuss your needs or ask any questions about the medicine.
Information from DengYueMed, China Drug Import and Export Wholesaler offers this information as a guide, not a substitute for professional medical advice. Consult your doctor before any treatment.




This text is worth everyone’s attention. How can I find out
more?
Thank you. Follow dengyuemed.com and we will update latest medical news more.