5 Powerful Trends Showing China Innovative Drugs Will Hit $100B+ by 2025
Table of Contents
By 2025, china innovative drugs transactions in China are forecast to exceed hundreds of billions USD. This article explores approval, licensing, and export acceleration—supported by partners such as HongKong DengYue Medicine, a top HongKong Pharmaceutical distributor, and robust China Medicine Wholesaler networks.
Introduction
China innovative drugs are on the cusp of a historic scaling. By 2025, it is widely projected that the transaction volume of these novel therapies in China will exceed hundreds of billions of US dollars. Alongside that rise, many domestic innovations are already approved overseas, licensing-out deals are surging, and export momentum is accelerating. This article explores the drivers behind this growth, the infrastructure and distribution partners enabling it, the risks to watch, and what stakeholders should do to position themselves for this wave of opportunity.
Forecasted Scale: Why Hundreds of Billions?
The projection that china innovative drugs will command a transaction market in the hundreds of billions by 2025 isn’t wishful thinking. Consider:
- China’s pharmaceutical market is one of the fastest-growing globally, and innovative therapies (oncology, biologics, gene therapies) command high unit prices.
- Licensing deals and cross-border partnerships are adding large sums to total transaction value.
- The shift from generic dominance to innovation increases average deal size per molecule.
- Policies such as priority reviews, national support for innovation, and favorable medical reimbursement coverage expand domestic market uptake.
As more first-in-class drugs reach the market and price premiums are accepted, transaction volume could well reach or surpass USD 100-300 billion territory.
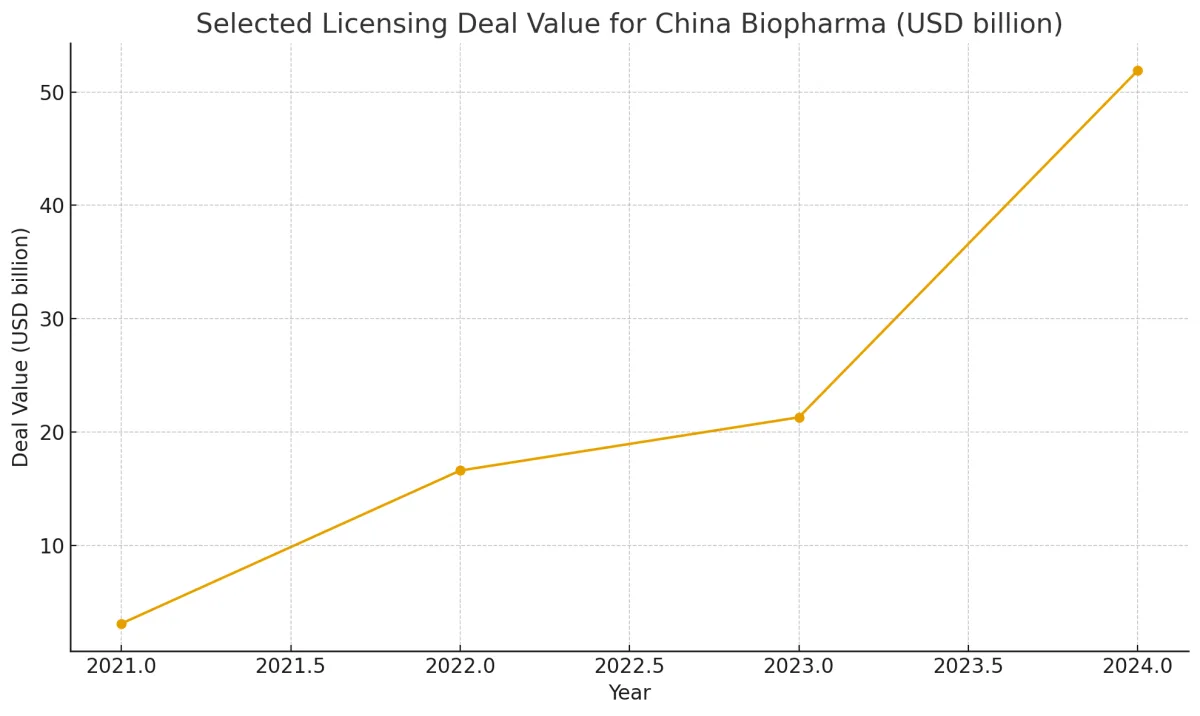
Overseas Approvals & Licensing Deals
One major sign of international integration: an increasing number of china innovative drugs have earned approval for sale in overseas markets. Chinese biotech firms are securing regulatory clearances from the U.S. FDA, European EMA, Japan PMDA, and other agencies. Alongside this, licensing out arrangements (where a domestic innovator grants rights to global companies) are multiplying. These deals often involve upfront payments, milestone payments, and royalties, significantly expanding the financial footprint of China’s innovation economy.
When licensing out, the value of transactions often includes global markets—not just domestic China—so the total market captured by china innovative drugs becomes far larger. This trend fuels optimism for that “hundreds of billions” forecast.
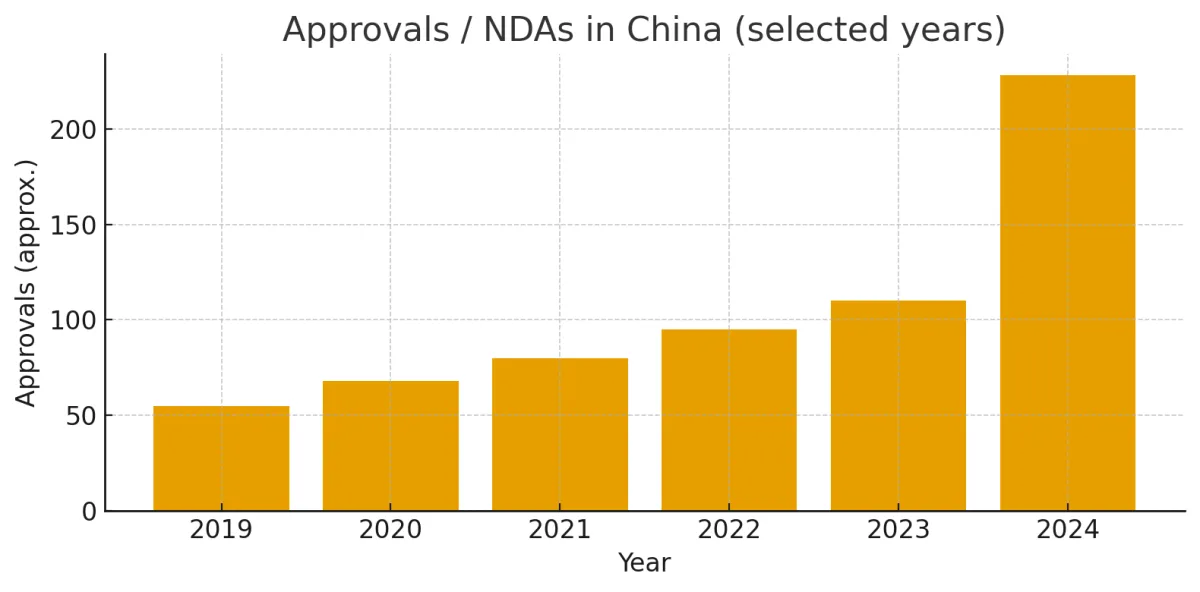
Accelerating Exports of Innovative Drugs
Export of innovative therapies is no longer a niche activity; it’s intensifying. Several factors drive this:
- Chinese firms develop export-grade formulations and packaging to comply with ICH/WHO standards.
- Exports of novel biologics and precision medicines are being shipped to markets in Southeast Asia, the Middle East, and even Europe.
- Institutions and governments abroad are increasingly open to adopting Chinese innovations, especially when cost and clinical data are favorable.
- Export deals often accompany licensing structures or co-development arrangements, further boosting transaction value.
Thus, china innovative drugs are not just for domestic consumption—they are becoming a global export narrative.
Distribution Infrastructure & Strategic Players
For china innovative drugs to reach global markets, robust distribution systems and strategic export partners are essential.
Hong Kong as a Trade Hub
Hong Kong plays a special role as a gateway between Mainland China and the world. With its efficient customs, strong legal framework, and logistical capacity, it becomes a natural base for re-export, value-added services, and compliance bridging.
Role of HongKong DengYue Medicine & HongKong Drug Wholesale Distributor
Exporters of china innovative drugs often partner with an experienced HongKong Drug Wholesale Distributor. HongKong DengYue Medicine, as a licensed HongKong Pharmaceutical distributor, is a standout example—providing full import/export licensing, cold-chain logistics, regulatory compliance, and traceability across the supply chain. Such a distributor serves as a reliable export node, facilitating global reach for domestic innovations.
Support from China Medicine Wholesaler Networks
On the Mainland side, China Medicine Wholesaler networks manage aggregation, warehousing, batch tracking, and domestic logistics. As these wholesalers upgrade to GDP standards and integrate digital traceability, they become capable partners in cross-border supply for china innovative drugs.
Together, these distribution layers – Mainland wholesalers + Hong Kong gateway distributors – form the backbone that allows the scale of export and transaction growth required for a multi-billion USD market.
Challenges & Risk Mitigation
While the outlook is exciting, several risks deserve attention:
- Regulatory divergence: Global markets have differing standards (safety, clinical data, IP). Some markets may demand additional trials or local bridging data.
- Intellectual property & data protection: Ensuring patents, data exclusivity, and trade secrecy is crucial.
- Quality consistency: Biologics, cell therapy, and advanced formulations are sensitive to manufacturing variation.
- Logistics and cold-chain breaks: Maintaining temperature control across long distances is expensive and risky.
- Reputational risk: Any adverse event or compliance breach can cast a shadow not just on one drug, but on the broader “china innovative drugs” brand.
Mitigation strategies include partnering with trusted exporters (like HongKong DengYue Medicine), investing in quality systems, auditing partners, building redundancy in logistics, and transparency in regulatory pledges.
Strategic Recommendations
To seize the 2025 opportunity for china innovative drugs:
- Innovators should focus on global readiness early—design trials with multiregional intent, meet ICH/WHO alignment.
- Distributors must build infrastructure for cold-chain, regulatory compliance, and traceability—especially China Medicine Wholesaler networks upgrading their systems.
- Export partners like HongKong Pharmaceutical distributor firms should package services around licensing, regulatory documentation, and global marketing support.
- Licensing strategies need to carefully balance control, royalty shares, and market access to maximize global value capture.
- Collaborate with credible gatekeepers: working with established HongKong Drug Wholesale Distributor firms (e.g. HongKong DengYue Medicine) mitigates risk and accelerates market entry.
Conclusion
By 2025, the transaction volume of china innovative drugs is set to surpass hundreds of billions of US dollars—driven by more overseas approvals, licensing out deals, and rapid export growth. But this transformation depends on the strength of distribution infrastructure, regulatory readiness, and trustworthy export partners. With support from China Medicine Wholesaler systems and strategic firms like HongKong DengYue Medicine, the vision of global Chinese innovation is within reach. For innovators, exporters, and institutional investors, aligning now with the right partners will determine who participates in this next phase of pharmaceutical leadership.