
ABSK061 Achondroplasia Orphan Drug Designation: Abbisko Therapeutics Advances Targeted Therapy
The ABSK061 achondroplasia orphan drug designation marks an important regulatory milestone in the development of targeted therapies for rare genetic skeletal disorders.
Developed by Abbisko Therapeutics, ABSK061 is a selective FGFR2/3 inhibitor designed to target the underlying molecular mechanism of achondroplasia rather than only managing symptoms.
Achondroplasia is a rare genetic bone growth disorder driven by mutations in the FGFR3 signaling pathway, leading to impaired skeletal development, particularly in pediatric patients. Despite ongoing research, treatment options that directly target disease biology remain limited.
The ABSK061 achondroplasia orphan drug designation therefore represents growing interest in mechanism-based therapies targeting FGFR signaling in rare bone diseases.
For Chinese pharmaceutical wholesalers, DengYueMed, this progress also highlights emerging opportunities in rare disease drug access and global distribution.
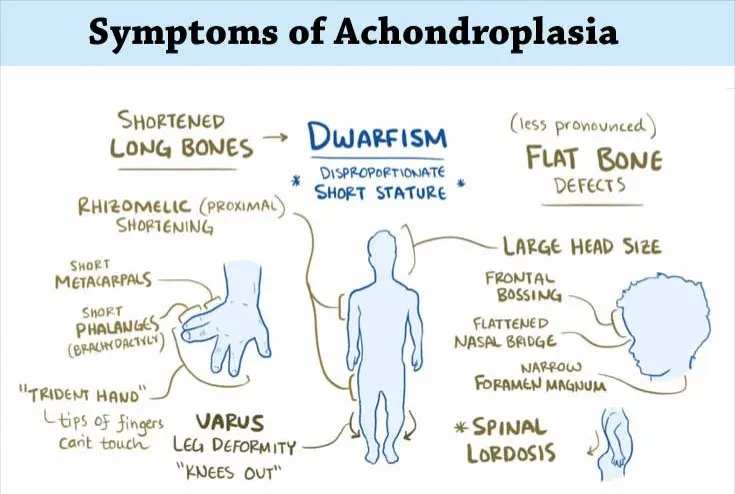
What Is Achondroplasia?
Achondroplasia is the most common form of dwarfism and is caused primarily by gain-of-function mutations in the FGFR3 gene.
This leads to:
- Overactivation of FGFR3 signaling
- Suppression of chondrocyte proliferation
- Impaired endochondral ossification
- Reduced long bone growth
The result is disproportionate short stature and skeletal abnormalities that become evident in early childhood.
FDA Orphan Drug Designation (ODD): Regulatory Incentives and Commercial Implications
On March 19, 2026, Abbisko Therapeutics announced that ABSK061 received FDA orphan drug designation for achondroplasia.
This ABSK061 achondroplasia orphan drug designation builds upon its earlier Rare Pediatric Disease Designation (RPDD), further strengthening its regulatory position in the United States and supporting accelerated development.
From a regulatory perspective, FDA orphan drug designation provides incentives that help accelerate rare disease drug development and support innovation in achondroplasia treatment.
Key benefits include: 👇
✅ Direct incentives:
- Tax credits for qualified clinical trial expenses
- Waiver of NDA/BLA application fees
- Up to 7 years of market exclusivity upon approval
✅ Indirect advantages:
- Increased interaction and guidance from the FDA
- Enhanced attractiveness for partnerships and licensing
- Reduced development risk for targeted therapies
For rare disease drugs, orphan drug designation often represents a critical transition from early development to commercial viability.
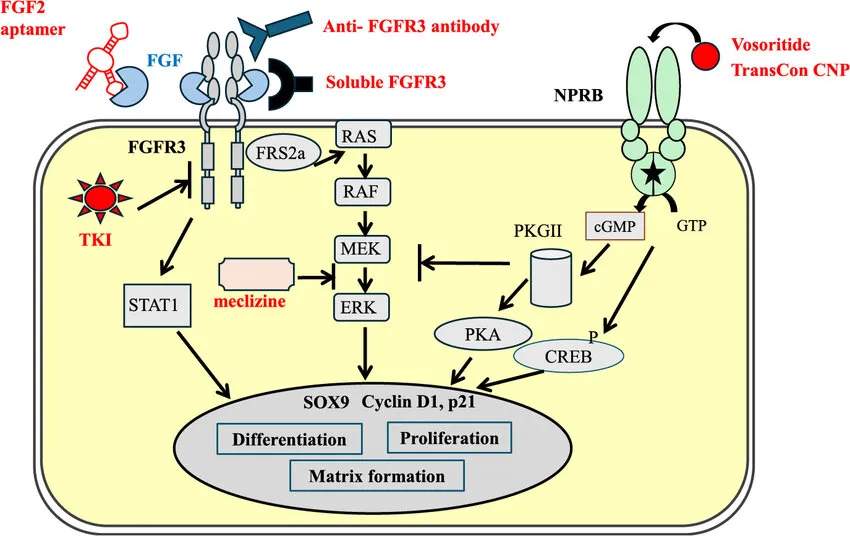
Disease Mechanism: FGFR3 Signaling and Skeletal Development
FGFR3 (fibroblast growth factor receptor 3) acts as a negative regulator of bone growth by controlling chondrocyte proliferation and differentiation. In achondroplasia, gain-of-function mutations in FGFR3 result in persistent activation of the receptor.
This abnormal signaling cascade involves:
- Ligand-independent activation of FGFR3
- Activation of downstream signaling pathways such as MAPK and STAT
- Suppression of chondrocyte proliferation and hypertrophic differentiation
As a result, endochondral ossification is impaired, leading to shortened limbs and restricted growth.
🎯 This mechanism is central to achondroplasia and provides a clear rationale for using FGFR inhibitors in achondroplasia treatment and supports the scientific basis behind the ABSK061 achondroplasia orphan drug designation.
Mechanism of ABSK061: Targeting FGFR Signaling in Achondroplasia
ABSK061 is designed to selectively inhibit FGFR2/3 kinase activity, directly targeting the signaling abnormalities associated with achondroplasia.
Its mechanism includes:
- Inhibition of FGFR3 phosphorylation
Reducing abnormal receptor activation - Downregulation of MAPK/ERK pathways
Supporting restoration of chondrocyte proliferation - Promotion of growth plate function
Potentially supporting cartilage development
This mechanism highlights the potential of ABSK061 as a targeted FGFR inhibitor for achondroplasia treatment, rather than a purely symptomatic therapy, reinforcing the importance of the ABSK061 achondroplasia orphan drug designation in targeted drug development.
Drug Profile and Differentiation: Small Molecule Advantages
Within the achondroplasia treatment landscape, ABSK061 represents a small molecule FGFR inhibitor with notable advantages:
1️⃣ High Selectivity for FGFR2/3
- Reduced off-target activity
- Lower risk of toxicity
- Improved long-term safety profile
2️⃣ Oral Administration in Pediatric Patients
Unlike injectable therapies, oral administration offers:
- Greater convenience for children and caregivers
- Reduced reliance on hospital-based care
- Improved adherence in long-term achondroplasia treatment
Route of administration plays a critical role in real-world outcomes for rare pediatric diseases.
3️⃣ Pharmacokinetics and Safety Potential
Preclinical studies suggest:
- Favorable oral bioavailability
- Stable drug exposure levels
- Predictable dose–response relationships
These features support continued development of ABSK061 as a targeted therapy for achondroplasia.
Clinical Development Strategy: Phase II Study in Pediatric ACH Patients
ABSK061 is currently being evaluated in a Phase II clinical trial in children aged 3–12 with achondroplasia. The first patient was dosed in China in December 2025.
As part of the development strategy following the ABSK061 achondroplasia orphan drug designation, key endpoints include:
- Annualized Growth Velocity (AGV)
- Height Z-score changes
- Bone age progression
Initial data are expected in the second half of 2026 and will guide further development, including potential Phase III studies and global expansion.
Competitive Landscape: Evolving Achondroplasia Treatment Strategies
The achondroplasia treatment landscape is rapidly evolving, with several therapeutic approaches under investigation:
1️⃣ CNP analogs – promoting bone growth
2️⃣ Anti-FGFR3 antibodies – blocking receptor activation
3️⃣ Small molecule FGFR inhibitors (e.g., ABSK061) – targeting signaling pathways
Among these, small molecule therapies like ABSK061 offer advantages in convenience and scalability, positioning them as a promising option in future achondroplasia treatment strategies.
Conclusion: ABSK061 and the Future of Achondroplasia Treatment
The ABSK061 achondroplasia orphan drug designation marks a significant milestone in the development of targeted therapies for rare skeletal disorders. It reflects regulatory recognition of the potential of FGFR inhibition as a strategy for achondroplasia treatment.
As clinical data continue to emerge, ABSK061 may become an important addition to the treatment landscape, offering a targeted and potentially more accessible option for patients.
At the same time, the advancement of therapies like ABSK061 is reshaping the global rare disease ecosystem. For HongKong DengYueMed, improving access to innovative treatments and supporting global distribution will remain a key strategic focus.
FAQ about ABSK061 Achondroplasia Orphan Drug Designation
What is ABSK061?
ABSK061 is a selective FGFR2/3 inhibitor in development for achondroplasia treatment. It targets abnormal FGFR3 signaling and has received FDA orphan drug designation for achondroplasia.
What is orphan drug designation?
Orphan drug designation is a status granted by the FDA to encourage the development of treatments for rare diseases by providing incentives such as tax benefits, fee waivers, and market exclusivity.
How does FGFR3 inhibition work in achondroplasia?
In achondroplasia, overactive FGFR3 signaling suppresses bone growth. FGFR3 inhibitors like ABSK061 block this pathway, reduce MAPK/ERK activation, and help support cartilage development and bone growth.
Why is ABSK061 considered a promising therapy for achondroplasia?
ABSK061 is a selective oral FGFR2/3 inhibitor with disease-targeted action. Combined with its FDA orphan drug designation for achondroplasia, it represents a mechanism-based approach that directly addresses disease biology rather than only symptoms.



