New Hope in Fighting Breast Cancer: Understanding the Targeted Drug Verzenio (Abemaciclib)
Verzenio has opened a new window of hope for many patients with HR-positive, HER2-negative breast cancer.
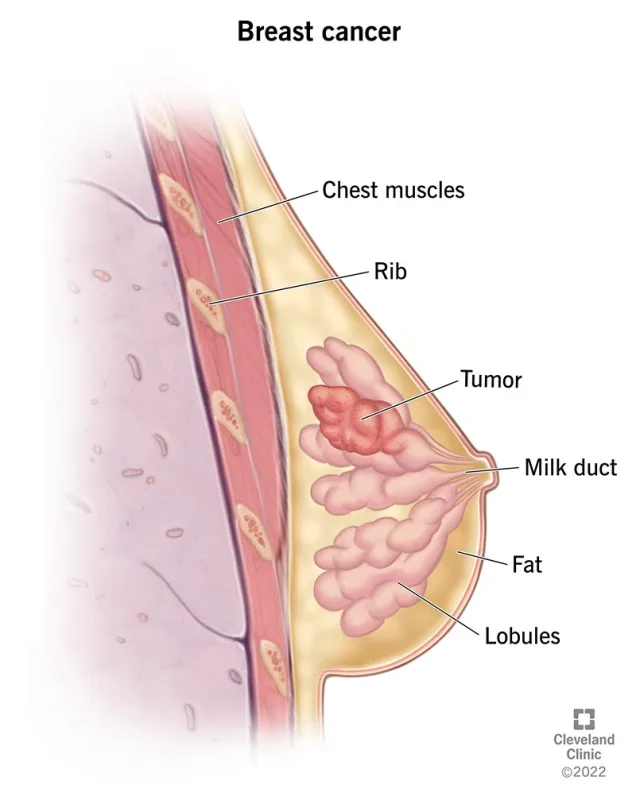
The Challenge of Breast Cancer
HR-positive, HER2-negative breast cancer, as the most common subtype, has long relied primarily on endocrine therapy (hormone therapy).
However, traditional treatments have their limitations.
For patients with metastatic hormone receptor-positive breast cancer, the response rate to first-line hormone therapy is only about 30%.
Another 30% of patients can maintain stable disease, but up to 40% of patients respond poorly to hormone therapy.
More concerning is that after first-line treatment fails, the response rate to second-line hormone therapy drops to below 10%, with a progression-free survival of only 3-4 months.

The CDK4/6 Inhibitor Revolution
In recent years, the focus of cancer treatment has gradually shifted from traditional chemotherapy to more targeted therapies.
Among these, the emergence of CDK4/6 inhibitors marks a significant breakthrough in the treatment of HR-positive, HER2-negative breast cancer.
So what are CDK4/6? These proteins act as “accelerator pedals” in the cell division cycle.
- Under normal circumstances, their activity is strictly regulated, but in cancer cells, this mechanism goes out of control, leading to unlimited cell division and proliferation.
- CDK4/6 inhibitors work by stepping on the brake of this “accelerator pedal,” preventing cancer cells from dividing.
What Makes Verzenio Unique
Verzenio has structural differences from the other two CDK4/6 inhibitors, giving it unique advantages.
In enzyme experiments, Verzenio demonstrated 14 times greater potency in inhibiting CDK4 and CDK6.
Verzenio works by preventing the phosphorylation of retinoblastoma protein, inducing cellular senescence and apoptosis, arresting cells in the G1 phase and preventing them from entering the S phase (DNA synthesis phase).
Verzenio is currently the only CDK4/6 inhibitor that doesn’t require a drug holiday, meaning patients can receive continuous treatment without periodic breaks.

Clinical Applications of Verzenio
Verzenio has received approval for multiple indications, providing treatment options for patients in different situations:
- In combination with aromatase inhibitors: As first-line hormone therapy for postmenopausal women with HR-positive, HER2-negative advanced or metastatic breast cancer
- In combination with fulvestrant: For treating women with HR-positive, HER2-negative advanced or metastatic breast cancer whose disease has progressed after endocrine therapy
- Monotherapy: For adult patients with HR-positive, HER2-negative advanced or metastatic breast cancer who have previously received endocrine therapy and chemotherapy after metastasis, and whose disease has progressed again
Clinical Evidence Support
The efficacy of Verzenio is strongly supported by evidence from multiple clinical trials.
The MONARCH 2 study showed that the Verzenio plus fulvestrant treatment group achieved a median progression-free survival of 16.4 months.
Significantly extending by 7.1 months compared to 9.3 months with fulvestrant alone.
The MONARCH 3 study, involving 493 postmenopausal women with HR-positive, HER2-negative advanced breast cancer, demonstrated that Verzenio combined with a non-steroidal aromatase inhibitor was significantly more effective than the control group.
Even more encouraging, the MONARCH E study confirmed that Verzenio combined with standard endocrine therapy for high-risk early breast cancer patients significantly reduced the risk of breast cancer recurrence by 28.7%.
Precautions and Side Effects
Like all anticancer drugs, Verzenio may also cause some side effects.
Diarrhea is the most common, with an incidence rate of up to 90%.
Other common side effects include:
- Fatigue (65%)
- Nausea (64%)
- Decreased appetite (45%)
- Abdominal pain (39%)
- Vomiting (35%)
- Reductions in various blood cell types
Patients should pay special attention to the fact that Verzenio may cause severe diarrhea that requires prompt treatment and management.
Regarding drug interactions, patients should avoid grapefruit products as they inhibit CYP3A4 enzymes, increasing the concentration of Verzenio in the blood and potentially leading to increased toxicity.

Verzenio (Abemaciclib) – Breast Cancer | HongKong DengYue Medicine
- Generic Name/Brand Name: Abemaciclib/Verzenio
- Indications: HR-positive / HER2-negative advanced/ metastatic breast cancer
- Dosage Form: Oral tablets
- Specification: 50 mg/100 mg/150 mg/200 mg × 14 tablets
Patient Care
For patients currently or about to receive Verzenio treatment, the following suggestions from HKdengyuemed may be helpful during the treatment process:
👉Strictly follow medical advice: Do not adjust the dose or stop taking the medication on your own.
👉Manage diarrhea: If loose stools occur, promptly inform your doctor.
👉Regular monitoring: Regular blood tests are needed during treatment.
👉Contraceptive measures: Effective contraception should be used during treatment and for 3 weeks after the last dose.
Scientific progress is gradually transforming breast cancer into a chronic, manageable disease, allowing more patients to regain hope and move toward the future.
FAQ about Understanding the Targeted Drug Verzenio (Abemaciclib)
Is Verzenio the same as chemo?
Verzenio (abemaciclib) isn’t chemotherapy. Instead, it’s a targeted therapy.
What type of treatment is Verzenio?
Verzenio is a brand of abemaciclib.
This medication treats certain types of breast cancer by stopping cancer cells from growing.
What are the long term effects of Verzenio?
So some of its side effects may be long-term.
For example, in clinical trials, nearly 70% of people taking Verzenio experienced anemia (low red blood cell level).
How long does Verzenio extend life?
In a clinical study, women taking Verzenio plus fulvestrant lived for a median of 46.7 months compared to 37.3 months on fulvestrant alone, regardless of menopausal status.