China Generic Drug Market 2026: Opportunities and Trends After Blockbuster Drug Patent Expiry
As global drug patents continue to expire, the China generic drug market is entering a new cycle of competition and opportunity.
Around 2026, the Chinese pharmaceutical industry is approaching an important milestone. Several globally important drugs are approaching patent expiration, including the widely discussed GLP-1 therapy Semaglutide.
✨ For Chinese pharmaceutical companies, this represents not only a significant commercial opportunity but also a signal that the China generic drug industry is transitioning from large-scale expansion toward stronger R&D capabilities and innovation.
From policy incentives and patent challenges to global market expansion, China’s generic drug sector is undergoing profound transformation.
As a pharmaceutical distributor in China, DengYueMed closely monitors developments in the China generic drug market and global pharmaceutical supply chains, providing international partners with drug information and pharmaceutical supply channels.
This article analyzes the major policy changes, market competition, and future trends shaping the China generic drug market in 2026.
Policy Upgrades Shaping the China Generic Drug Market and Generic Drug Development in China
In January 2026, China’s National Health Commission released the Fourth Batch of the Encouraged Generic Drug Catalogue, which includes 21 drug products and 47 specifications.
Compared with previous versions, the updated catalog reflects several important changes in China’s pharmaceutical policy.
1 Addressing Key Technology Gaps in China Generic Drug Development
The policy increasingly focuses on therapeutic areas where domestic R&D capabilities remain relatively limited, such as:
- Drugs with novel mechanisms of action
- Radiopharmaceuticals
- Complex formulations
For example, Difelikefalin, a treatment for pruritus in dialysis patients with chronic kidney disease, was included in the encouraged generic drug list.
This highlights China’s efforts to expand its capabilities in high-value generic drug development.
2 Greater Emphasis on International Clinical Value
Some drugs included in the catalog have already been recommended by international clinical guidelines.
One example is Suvorexant, an orexin receptor antagonist used for the treatment of insomnia.
This indicates that China’s policy direction is shifting from simply addressing drug shortages toward improving treatment quality and clinical value.
3 Increasing Focus on Patient Experience
Drugs used in assisted reproductive therapy were also included in the catalog, such as:
- Progesterone vaginal sustained-release gel
- Progesterone vaginal suppositories
These dosage forms can reduce the discomfort associated with injectable drugs and improve patient adherence.
Competitive Landscape of the China Generic Drug Market in Early 2026
Rising Number of First Generic Drug Approvals in China
In January 2026, China approved eight first generic drugs, representing one of the highest monthly numbers in recent years.
These products include:
- Ivocacine tablets
- Bromhexine hydrochloride inhalation solution
- Azilsartan amlodipine tablets
- Brexpiprazole orally disintegrating tablets
| Generic Name | Dosage Form | Marketing Authorization Holder (MAH) | Approval Date | Therapeutic Category |
|---|---|---|---|---|
| Bromhexine Hydrochloride | Inhalation Solution | Hefei Guoke Nuoke Pharmaceutical Co., Ltd. | 2026/1/5 | Respiratory System Drugs |
| Pyridostigmine Bromide | Oral Solution | Shanghai Zhongxi Pharmaceutical Co., Ltd. | 2026/1/5 | Nervous System Drugs |
| Sodium Bicarbonate Hemofiltration Replacement Solution | Hemofiltration Replacement Solution / CRRT Replacement Solution | Shanghai Fenglin Biotechnology Co., Ltd. | 2026/1/5 | Blood System / Nephrology |
| Evocalcet | Tablets | Jiangxi Kexin Pharmaceutical Co., Ltd. | 2026/1/14 | Endocrine & Metabolic Drugs |
| Amlodipine + Azilsartan (Compound Preparation) | Tablets | Shanghai Yiren Pharmaceutical Co., Ltd. | 2026/1/14 | Cardiovascular Drugs |
| Levornidazole Disodium Phosphate | Lyophilized Powder for Injection | Huaren Pharmaceutical Co., Ltd. | 2026/1/26 | Anti-infective Drugs |
| Brexpiprazole | Tablets | Qilu Pharmaceutical Co., Ltd. | 2026/1/27 | Psychiatric Drugs |
| Cefditoren Pivoxil | Tablets | Fuan Pharmaceutical Group Qingyutang Pharmaceutical Co., Ltd. | 2026/1/30 | Anti-infective Drugs |
This trend suggests that Chinese pharmaceutical companies are rapidly strengthening their capabilities in generic drug R&D, regulatory submission, and clinical research.
Most Competitive Targets in the China Generic Drug Pipeline
According to regulatory filing data, several drugs have become highly competitive targets for generic development, including:
- Finerenone
- Dapagliflozin
- Azilsartan
For some products, five to six pharmaceutical companies have submitted marketing applications, indicating intense competition in the future China generic drug market.
Patent Expiration of Blockbuster Drugs Driving China Generic Drug Competition
Upadacitinib: A Multi-Billion-Dollar Drug Facing Generic Competition
Upadacitinib is a JAK inhibitor developed by AbbVie and is widely used to treat:
- Rheumatoid arthritis
- Atopic dermatitis
- Ulcerative colitis
Global sales approached nearly $6 billion in 2024.

Currently, more than 30 Chinese pharmaceutical companies are involved in generic development, and several products are expected to enter the market in the coming years.
Abemaciclib: Patent Competition in Oncology Drugs
Abemaciclib is a CDK4/6 inhibitor developed by Eli Lilly for the treatment of breast cancer.
Global sales have already exceeded $5 billion.

In China, only two companies have currently submitted generic applications, meaning the competitive landscape remains relatively concentrated.
China’s Innovative Drugs Are Also Entering the Generic Cycle
Another noteworthy trend is that Chinese-developed innovative drugs are also beginning to face generic competition.
For example, Edaravone Dexborneol, developed in China, is used to treat neurological damage caused by acute ischemic stroke.
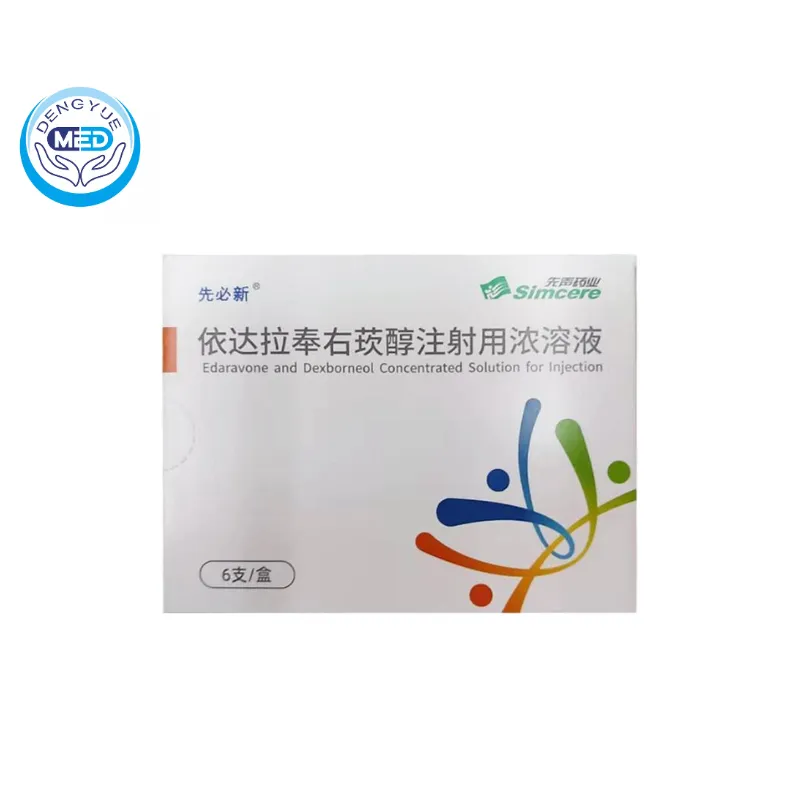
Although the drug was launched only a few years ago, multiple companies have already submitted generic applications.
This indicates that the Chinese pharmaceutical industry is gradually shifting from imitating foreign drugs to a dual model of innovation and generics.
GLP-1 Patent Expiration and the Future GLP-1 Biosimilar Market in China
Semaglutide Patent Expiration in China
One of the most closely watched events in the China pharmaceutical industry in 2026 is the expiration of the core compound patent for Semaglutide.
According to publicly available patent information, the Chinese core compound patent CN101133082B is expected to expire on March 20, 2026.
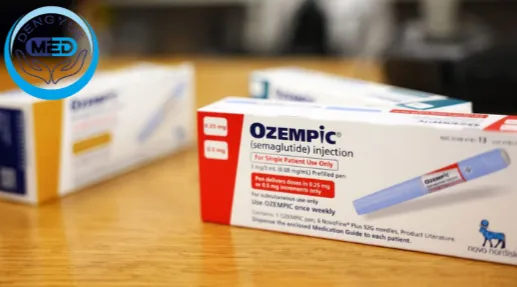
The patent is held by Novo Nordisk and represents a key piece of intellectual property behind the drug’s global commercialization.
Semaglutide is one of the best-selling GLP-1 therapies in the world and is widely used for:
- Type 2 diabetes treatment
- Obesity management
Global sales exceeded $20 billion in 2024.
Chinese Companies Accelerate GLP-1 Biosimilar Development
Currently, multiple Chinese pharmaceutical companies are developing Semaglutide biosimilars or related GLP-1 products, including:
- Lizhu Pharmaceutical
- Huadong Medicine
- Qilu Pharmaceutical
- Chia Tai Tianqing
Some candidates have already entered late-stage clinical trials or regulatory submission stages.
As biosimilars enter the market, prices may decline significantly, improving global drug accessibility.
Capability Upgrades Among Chinese Generic Drug Manufacturers
As competition intensifies, Chinese generic drug manufacturers are adopting different strategies to strengthen competitiveness.
✅ Specialization in Therapeutic Segments
Some companies are focusing on specific therapeutic areas, such as:
- CRRT replacement fluids
- Inhalation formulations
- Complex dosage forms
This niche specialization strategy is becoming increasingly common.
✅ Stronger Patent Challenge Strategies
More Chinese pharmaceutical companies are actively challenging originator drug patents through:
- Patent invalidation applications
- Patent design-around strategies
This approach allows companies to enter the market earlier and obtain first-generic advantages.
✅ Improved Formulations and Modified Drugs
Beyond simple generics, companies are also developing:
- New dosage forms
- Modified formulations
- New indications
This strategy can increase the clinical value of drugs while extending product life cycles.

Future Trends in the China Generic Drug Market and Global Pharmaceutical Supply Chain
Looking ahead, several trends are likely to shape the future of the China generic drug market.
1 Parallel Development of Generics and Innovation
Many companies are using generic drug development to accumulate technical expertise before moving into innovative drug R&D.
2 Expansion into Global Pharmaceutical Markets
As domestic competition intensifies, Chinese pharmaceutical companies are expanding into emerging markets such as:
- Southeast Asia
- The Middle East
- Latin America
- Africa
3 Digitalization and Online Pharmaceutical Distribution
In China, some medications are already being distributed through regulated online pharmacy platforms.
This digital model may continue to expand globally as pharmaceutical supply chains evolve.
Conclusion: The Future of the China Generic Drug Market After Patent Expirations
The year 2026 may represent a turning point for the China generic drug industry.
From regulatory policy and R&D capability to global market participation, Chinese pharmaceutical companies are steadily strengthening their role in the global pharmaceutical supply chain.
As more blockbuster drugs approach patent expiration, the China generic drug market will play an increasingly important role in improving drug accessibility and reducing treatment costs worldwide.
With the continuous development of China’s generic drug industry, this market is attracting increasing attention from international healthcare institutions and pharmaceutical buyers.
DengYueMed will continue to monitor developments in the China generic drug market and provide global partners with pharmaceutical information and supply support.
FAQ about China Generic Drug Market
Does China produce generic drugs?
Yes. China produces a large number of generic drugs and is one of the world’s major pharmaceutical manufacturing hubs. Chinese companies typically develop and manufacture generics after the patents of original drugs expire, helping improve access to more affordable medicines worldwide.
What is the role of China in the global generic drug API market?
China plays a key role in the global generic drug API market and is one of the largest suppliers of pharmaceutical raw materials. It produces a significant share of active pharmaceutical ingredients (APIs), key starting materials (KSMs), and related chemicals used in generic drug manufacturing worldwide.
How big is the generic drug market?
The global generic drug market is large and continues to grow steadily. It was valued at about $468 billion in 2025 and is projected to reach around $762 billion by 2035, expanding at an average annual growth rate of roughly 5% over the next decade.
What is the most common drug used in China?
There is no single most commonly used drug in China, as usage varies by disease and region. However, widely used medications include antibiotics, cardiovascular drugs, and treatments for chronic conditions such as hypertension and diabetes.