Bispecific ADC Development in Breast Cancer: Emerging Targets and Clinical Progress
Breast cancer is the most common malignancy among women worldwide, with incidence continuing to rise. Precision molecular subtyping and individualized therapies have improved outcomes, but tumor heterogeneity, antigen escape, and drug resistance remain major challenges.
Antibody–drug conjugates (ADCs) enable targeted delivery of cytotoxic payloads, improving efficacy while reducing systemic toxicity. However, traditional ADCs face limitations related to antigen dependence and resistance.
✨ Bispecific antibody–drug conjugates (BsADCs) address these issues by simultaneously targeting two tumor-associated antigens, enhancing internalization, payload delivery, and bystander killing.
With advances in antibody engineering and payload design, bispecific ADC development in breast cancer has become a key research focus.
In this article, DengYueMed reviews recent progress in bispecific ADC development in breast cancer and summarizes its potential therapeutic implications.
Biological Rationale for Bispecific ADC Development in Breast Cancer
Breast cancer is a heterogeneous disease characterized by diverse molecular subtypes, including hormone receptor–positive, HER2-positive, and triple-negative breast cancer (TNBC). Differences in tumor biology and antigen expression often limit the efficacy of single-target therapies such as conventional antibody–drug conjugates (ADCs).
Tumor-associated antigens such as HER2, TROP2, EGFR, and HER3 can vary across tumor cells and metastatic sites, contributing to treatment resistance.
This heterogeneity provides an important rationale for bispecific ADC development in breast cancer, as BsADCs can simultaneously target two tumor antigens and improve tumor cell recognition and payload delivery.
In addition, interactions within the ERBB signaling network, particularly HER2–HER3 heterodimers, play a key role in tumor growth and survival, further supporting bispecific ADC development in breast cancer as a promising therapeutic strategy.
Structure and Design Principles of BsADCs
BsADCs consist of three core components: a bispecific antibody, a linker, and a cytotoxic payload. After binding to antigens on the tumor cell surface, the BsADC undergoes receptor-mediated endocytosis and enters the cell.
The payload is subsequently released in the endolysosomal compartment through linker cleavage, enabling precise tumor cell killing.
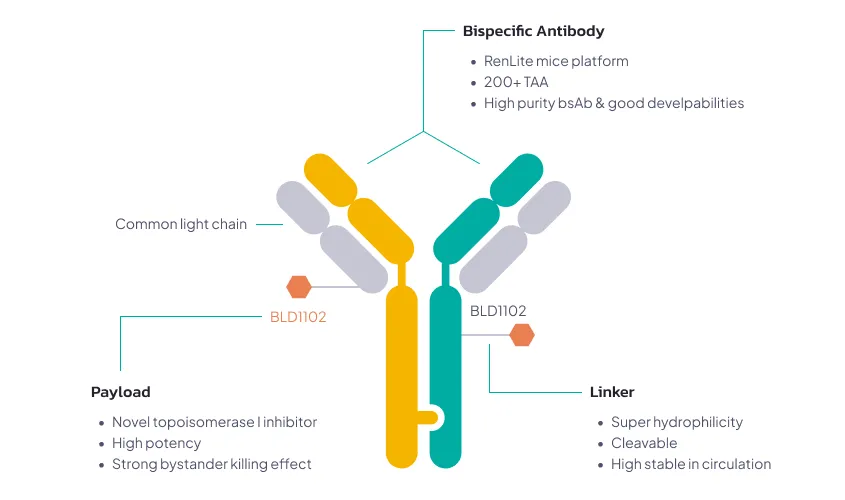
The development of BsADCs requires careful consideration of several key factors:
- Target antigen biology
Two target antigens should ideally be co-expressed on the same tumor cell to ensure effective dual targeting. - Antibody specificity
Highly selective antigen binding helps minimize off-target toxicity. - Payload cytotoxicity
The payload should effectively kill cancer cells and ideally possess membrane permeability to enable a bystander effect. - Linker stability and cleavability
The linker must remain stable in circulation but release the payload efficiently inside tumor cells. - Conjugation site
Conjugation strategy determines ADC homogeneity and the drug-to-antibody ratio (DAR), which significantly affects both safety and efficacy.
A major advantage of BsADCs is the bystander effect. Membrane-permeable payloads released in dual-positive cells can diffuse to neighboring tumor cells with single or no antigen expression, helping overcome tumor heterogeneity.
This characteristic is particularly important in bispecific ADC development in breast cancer, where antigen heterogeneity is common.
Advances in HER2-Targeted BsADC Research
HER2-positive breast cancer accounts for approximately 15–20% of all breast cancer cases. Therapeutic strategies have evolved from monoclonal antibodies to conventional ADCs. However, heterogeneous HER2 expression and resistance to receptor internalization continue to limit therapeutic outcomes.
BsADCs provide a novel strategy to address low HER2 expression and drug resistance through dual-epitope binding and enhanced receptor clustering. As a result, bispecific ADC development in breast cancer has increasingly focused on HER2-based designs.
Dual-Epitope HER2 BsADCs
ZW49
ZW49 conjugates an auristatin payload to the bispecific anti-HER2 antibody ZW25 (zanidatamab) via a cleavable linker.
Preclinical studies demonstrated potent antitumor activity and favorable tolerability. Early phase I clinical data showed:
- Objective response rate (ORR): 13%
- Main adverse event: grade 1 keratitis
- No severe pulmonary toxicity or treatment-related deaths observed
MEDI4276
MEDI4276 combines trastuzumab with the 39S antibody in a dual-epitope configuration and carries the microtubule inhibitor tubulysin derivative AZ13599185.
Phase I trials demonstrated limited clinical activity, and doses above 0.3 mg/kg resulted in unacceptable toxicity, which restricted further development.
JSKN003
JSKN003 utilizes the KN026 dual-epitope HER2 antibody with site-specific conjugation of a DXd payload.
Phase I data demonstrated:
- Disease control rate (DCR): 90.6%
- Favorable tolerability
- No dose-limiting toxicities observed
JSKN003 has now advanced into phase III clinical trials and represents a significant milestone in bispecific ADC development in breast cancer.
Dual-Antigen HER2 BsADCs
Another strategy involves targeting HER2 together with rapidly internalizing receptors such as CD63, PRLR, or APLP2. Preclinical studies suggest that these combinations may enhance receptor internalization and lysosomal trafficking, improving payload delivery efficiency while reducing dependence on HER2 expression levels.
Examples include:
- HER2 × CD63: enhances delivery to late endosome–lysosome compartments
- HER2 × PRLR: utilizes PRLR internalization to promote HER2 degradation and improve payload transport
- HER2 × APLP2: facilitates HER2 internalization and optimizes lysosomal trafficking
HER2/HER3 BsADCs
The HER2/HER3 heterodimer is an important oncogenic signaling unit in breast cancer. HER2/HER3 BsADCs have demonstrated potent and selective antitumor activity in both cell lines and animal models, with an average DAR of approximately 2.89.
Overall, bispecific ADC development in breast cancer targeting HER2 mainly follows two technological strategies: 👇
1️⃣ Dual-epitope HER2 targeting to enhance receptor clustering and internalization
2️⃣ Dual-antigen targeting involving HER2 and other rapidly internalizing receptors to improve payload delivery efficiency
EGFR × HER3-Targeted BsADCs
EGFR (HER1) and HER3 are key members of the ERBB receptor family and play critical roles in epithelial tumor proliferation, survival, and migration.
Single-target EGFR antibodies or tyrosine kinase inhibitors (TKIs) often lose efficacy due to resistance mechanisms such as sensitizing mutations, receptor upregulation, or activation of bypass signaling pathways.
Bispecific ADC development in breast cancer has therefore explored EGFR/HER3 dual targeting as a strategy to overcome these resistance mechanisms.
💊 BL-B01D1 is an EGFR/HER3 BsADC that conjugates the topoisomerase I inhibitor ED04 to a bispecific antibody with an average DAR of approximately 8.
A phase I study in locally advanced or metastatic HER2-negative breast cancer (IHC 0–2+/ISH−) evaluated BL-B01D1 at 2.5 mg/kg every three weeks.
Results showed:
- ORR: 36.4%
- DCR: 80.2%
- Median progression-free survival (PFS): 6.9 months
- Duration of response (DoR): 9.7 months
- Treatment-related adverse events (TRAEs) were manageable, mainly hematologic and gastrointestinal toxicities
- Discontinuation rate: 5.0%
- No cardiotoxicity, cytokine release syndrome, or interstitial lung disease observed
Mechanistically, EGFR × HER3 BsADCs simultaneously inhibit ERBB signaling pathways, enhance receptor-mediated internalization, and improve payload delivery efficiency, thereby helping overcome resistance observed with single-target EGFR therapies.
These results highlight the growing importance of bispecific ADC development in breast cancer targeting ERBB family receptors.
TROP2 × HER3-Targeted BsADCs
Trop2 is highly expressed in multiple solid tumors, particularly triple-negative breast cancer (TNBC), making it an attractive therapeutic target.
TROP2/HER3 BsADCs enhance treatment selectivity and cytotoxic potency while addressing tumor heterogeneity.
💊 JSKN016 uses a cleavable linker to conjugate a topoisomerase I inhibitor payload with a DAR of 4.
Phase I results showed:
- ORR: 80% (4 of 5 mTNBC patients achieved partial response)
- TRAEs mainly included nausea, vomiting, and stomatitis
- Grade ≥3 adverse events: 10.5%
- Good overall tolerability
- No cardiotoxicity or treatment-related interstitial lung disease observed
- Maximum tolerated dose not yet reached
Mechanistically, the therapy works through:
- selective killing of dual-positive tumor cells
- enhanced internalization and payload release
- bystander killing that helps overcome tumor heterogeneity
These findings further support the therapeutic potential of bispecific ADC development in breast cancer, particularly for aggressive TNBC subtypes.
Overview of BsADC Candidates in Breast Cancer
With continuous progress in bispecific antibody engineering, linker chemistry, and cytotoxic payload design, an increasing number of BsADCs are being developed for breast cancer therapy.
Current bispecific ADC development in breast cancer mainly focuses on two design strategies:
- dual-epitope HER2 targeting
- dual-antigen combinations involving HER2, EGFR, or TROP2
Several BsADC candidates have already entered clinical development and demonstrated promising antitumor activity across different breast cancer subtypes.
Table 1. Representative BsADC candidates in clinical development for breast cancer
| Drug | Target | Payload | DAR | Clinical Phase | Key Features |
|---|---|---|---|---|---|
| ZW49 | HER2 dual epitope | Auristatin toxin | ~2 | Phase I | Based on bispecific anti-HER2 antibody ZW25 (zanidatamab), enhances receptor clustering and internalization |
| MEDI4276 | HER2 dual epitope | Tubulysin derivative (AZ13599185) | ~4 | Phase I | Dual-epitope design improves internalization but development limited by toxicity |
| JSKN003 | HER2 dual epitope | DXd (topoisomerase I inhibitor) | ~4 | Phase III | Site-specific conjugation with high disease control rate |
| BL-B01D1 | EGFR × HER3 | ED04 (topoisomerase I inhibitor) | ~8 | Phase I | High DAR design improves cytotoxic payload delivery |
| JSKN016 | TROP2 × HER3 | Topoisomerase I inhibitor | ~4 | Phase I | Designed for TNBC with strong bystander effect |
Overall, bispecific ADC development in breast cancer is becoming increasingly diversified in terms of target combinations while continuously optimizing payload selection, DAR control, and conjugation strategies.
👩🔬 These technological advances provide an important foundation for improving treatment selectivity, expanding indications, and enhancing safety.
Innovations and Challenges of BsADCs in Breast Cancer Therapy
Bispecific antibody–drug conjugates (BsADCs) represent an emerging strategy in breast cancer therapy with several key advantages.
✅ Improved selectivity and internalization
Dual-epitope or dual-target binding enables more precise tumor targeting while enhancing receptor-mediated internalization and payload delivery.
✅ Expanded therapeutic coverage
BsADCs may benefit patients with limited treatment options, including HER2-low, HER2-negative, and triple-negative breast cancer (TNBC).
✅ Balanced efficacy and safety
Technologies such as site-specific conjugation, cleavable linkers, and the bystander effect may improve the therapeutic window and reduce off-target toxicity.
Despite these advantages, several challenges remain, including toxicity management, complex engineering and manufacturing costs, and scientific issues such as immunogenicity, tumor heterogeneity, linker stability, and payload release control.
Future bispecific ADC development in breast cancer will likely focus on optimizing target combinations, improving linker and payload design, developing biomarker-driven patient selection, and enhancing manufacturing consistency. 🚀
Conclusion
Bispecific antibody–drug conjugates (BsADCs) are leading a new wave of innovation in breast cancer therapy. Their dual-targeting design enhances therapeutic efficacy and selectivity while offering new hope for patients with low HER2 expression and triple-negative breast cancer.
With continued advances in target optimization, payload and linker engineering, and biomarker-driven precision medicine, bispecific ADC development in breast cancer is expected to achieve a better balance between efficacy and tolerability in clinical practice.
Within the global clinical trial and pharmaceutical supply ecosystem, HongKong DengYue Medicine supports the information exchange and accessibility of BsADCs and other innovative therapeutics, helping promote their research development and clinical availability across different regions.
FAQ about Bispecific ADC Development in Breast Cancer
What are the approved ADCs for breast cancer?
Several antibody–drug conjugates (ADCs) are approved for breast cancer treatment, including Trastuzumab emtansine, Trastuzumab deruxtecan, and Sacituzumab govitecan. These ADCs target specific tumor antigens such as HER2 or TROP2 to deliver cytotoxic drugs directly to cancer cells.
What is the difference between ADC and bispecific?
Antibody–drug conjugates (ADCs), such as Trastuzumab deruxtecan, use a single antibody to target one antigen and deliver a cytotoxic drug to cancer cells. In contrast, bispecific antibodies are engineered to bind two different targets simultaneously, potentially enhancing tumor targeting or therapeutic activity.
What is the new drug for breast cancer in 2026?
A notable new drug for breast cancer is Datopotamab deruxtecan, a TROP2-targeted antibody–drug conjugate approved for previously treated HR-positive, HER2-negative metastatic breast cancer. New indications and combinations of Trastuzumab deruxtecan have also expanded treatment options for HER2-positive and HER2-low breast cancer.
Which breast cancer patients could benefit from bispecific ADC therapy?
BsADCs may benefit patients with HER2-low, HER2-negative, or triple-negative breast cancer (TNBC), as they can overcome antigen heterogeneity and resistance mechanisms that limit traditional therapies. By targeting multiple antigens, BsADCs broaden treatment coverage and improve therapeutic precision.