Approved Cell and Gene Therapy Drugs in China and Key Differences Between Cell Therapy and Gene Therapy
The landscape of approved cell and gene therapy drugs in China is rapidly evolving, as these advanced treatment approaches are increasingly recognized worldwide for their revolutionary potential in treating cancer, genetic disorders, and rare diseases.
Currently, China hosts more than 200 clinical trials related to cell and gene therapy, including a large number of CAR-T clinical studies. Globally, China is widely regarded as the second-largest CGT research hub after the United States, and it also has one of the largest numbers of CAR-T clinical studies worldwide. 🚀
Compared with the rapid expansion of research activity, the number of products that have received regulatory approval remains relatively limited. At present, nine approved cell and gene therapy drugs in China have received regulatory approval, including: 🔽
- 7 CAR-T cell therapies
- 1 stem cell therapy
- 1 gene therapy
These products are primarily used to treat relapsed or refractory hematologic malignancies, immune-related diseases, and genetic disorders.
Hong Kong DengYueMed provides an overview of approved cell and gene therapy drugs in China, including their technological principles, key differences, and currently approved products.
What Is Cell Therapy?
Cell therapy refers to a treatment approach in which living cells are processed or genetically modified and then infused into a patient to treat disease. These cells may originate from the patient (autologous cells) or from a donor (allogeneic cells).
Cell therapy typically works through several mechanisms:
- Immune attack on tumors: genetically engineered immune cells recognize and destroy cancer cells
- Tissue repair and regeneration: stem cells promote recovery of damaged tissues
- Immune regulation: modulating immune responses in autoimmune or inflammatory diseases
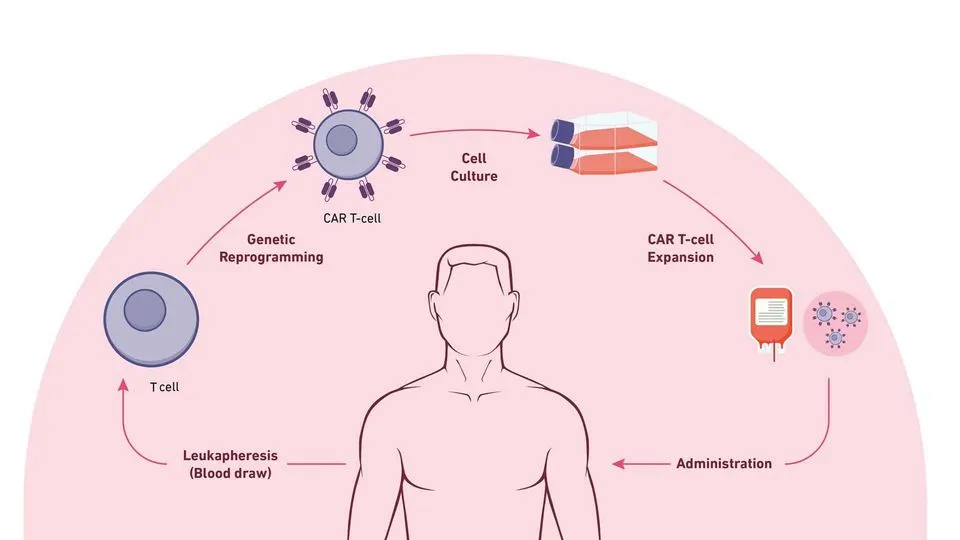
✨ The most mature form of cell therapy currently used in clinical practice is CAR-T cell therapy. Several CAR-T products have already become approved cell and gene therapy drugs in China, representing major progress in advanced cancer immunotherapy.
This therapy introduces chimeric antigen receptor (CAR) genes into a patient’s T cells using viral vectors or other gene engineering techniques, enabling them to recognize specific tumor antigens and eliminate cancer cells.
For example, in the treatment of multiple myeloma, CAR-T cells are engineered to recognize the B-cell maturation antigen (BCMA) antigen, allowing them to identify and destroy malignant plasma cells.
Most approved cell therapies worldwide are used to treat hematologic malignancies, including:
- Diffuse large B-cell lymphoma (DLBCL)
- Multiple myeloma
- B-cell acute lymphoblastic leukemia (B-ALL)
What Is Gene Therapy?
Gene therapy is a technology that treats disease by modifying or supplementing genetic information within human cells.
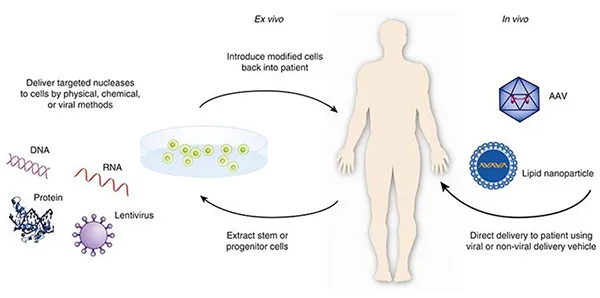
Unlike cell therapy, the core goal of gene therapy is to repair or regulate gene expression. Common approaches include:
1️⃣ Gene Replacement
Introducing a functional gene to replace a defective one.
2️⃣ Gene Silencing
Suppressing the expression of harmful genes.
3️⃣ Gene Editing
Directly modifying DNA sequences using advanced gene-editing technologies.
Most gene therapies use viral vectors as delivery systems, such as:
- AAV (Adeno-Associated Virus)
- Lentiviral vectors
These vectors deliver therapeutic genes into target cells, enabling long-term expression of functional proteins and restoration of cellular function.
Gene therapy is commonly applied to treat genetic diseases, rare diseases, and certain cancers.
In recent years, gene therapy technologies have also been integrated with cell therapy. For example, CAR-T therapy itself relies on gene engineering to modify T cells.
Key Differences in the Landscape of Cell Therapy vs Gene Therapy in China
Although both belong to the category of advanced therapies, they differ significantly in therapeutic logic and technological approach.
| Dimension | Cell Therapy | Gene Therapy |
|---|---|---|
| Therapeutic target | Living cells | Genes |
| Core mechanism | Cells perform therapeutic functions | Modify or supplement genes |
| Common technologies | CAR-T, stem cell therapy | AAV gene therapy, gene editing |
| Main indications | Hematologic malignancies, immune diseases | Genetic disorders, rare diseases |
| Treatment characteristics | Personalized cellular therapy | Long-term gene expression |
From a technological perspective, some therapies involve both approaches. For example, CAR-T therapy relies on gene engineering to modify T cells, meaning it incorporates elements of both cell therapy and gene therapy.
👉 This integration is also reflected in several approved cell and gene therapy drugs in China, many of which are based on genetically engineered immune cells.
Approved Cell Therapy Drugs in China
Several therapies represent the most important approved cell and gene therapy drugs in China, primarily including CAR-T therapies and stem cell therapies.
1. Axicabtagene Ciloleucel (Yescarta)
Developer: Fosun Kite Biotechnology
Approval Date: June 2021
Indication: Relapsed or refractory large B-cell lymphoma after ≥2 lines of therapy
Clinical Value:
Axicabtagene ciloleucel was the first CAR-T therapy approved in China, providing a new immunotherapy option for lymphoma patients who have failed conventional treatments.
2. Relmacabtagene Autoleucel (Carteyva)
Developer: JW Therapeutics
Approval Date: September 2021
Indications:
- Relapsed or refractory large B-cell lymphoma
- Relapsed or refractory B-cell acute lymphoblastic leukemia in patients ≤25 years old
Clinical Value:
Relmacabtagene autoleucel is one of the key domestically developed CAR-T products in China, demonstrating high response rates in relapsed and refractory patients.
3. Equecabtagene Autoleucel (FuKeSu)
Developer: IASO Biotherapeutics / Innovent Biologics
Approval Date: June 2023
Indication: Relapsed or refractory multiple myeloma after ≥3 lines of therapy
Clinical Value:
By targeting BCMA, equecabtagene autoleucel provides a new treatment option for multiple myeloma patients who have failed multiple prior therapies.

4. Inaticabtagene Autoleucel (YuanRuiDa)
Developer: Hrain Biotechnology
Approval Date: November 2023
Indication: Adult relapsed or refractory B-cell acute lymphoblastic leukemia
Clinical Value:
Inaticabtagene autoleucel represents an important CAR-T treatment option for adult B-ALL patients in China.
5. Zevorcabtagene Autoleucel (SaiKaiZe)
Developer: CARsgen Therapeutics
Approval Date: March 2024
Indication: Relapsed or refractory multiple myeloma after at least three prior therapies including a proteasome inhibitor and an immunomodulatory agent
Clinical Value:
Zevorcabtagene autoleucel further expands the BCMA CAR-T treatment landscape in China.

6. Ciltacabtagene Autoleucel (Carvykti)
Developer: Legend Biotech
Approval Date: August 2024
Indication: Relapsed or refractory multiple myeloma
Clinical Value:
Ciltacabtagene autoleucel has demonstrated deep and durable responses in multiple myeloma patients.
7. Ranicabtagene Autoleucel (HICARA)
Developer: Hrain Biotechnology
Approval Date: July 2025
Indication: Relapsed or refractory large B-cell lymphoma
Clinical Value:
Ranicabtagene autoleucel represents China’s first fully domestically developed CD19 CAR-T product for r/r LBCL.
8. Amimatoside Injection (RuiBoSheng)
Developer: Pluristyx Biotech
Approval Date: January 2025 (Conditional Approval)
Indication: Acute graft-versus-host disease (aGVHD) after failure of second-line or later therapies
Clinical Value:
Amimatoside is China’s first approved stem cell therapy, utilizing mesenchymal stem cells (MSCs) to regulate immune responses and reduce inflammatory damage caused by graft-versus-host disease.
Approved Gene Therapy Drug in China
In China, gene therapy is gradually moving from research into clinical application. With advances in gene delivery technologies and viral vector engineering, gene therapy products have begun to enter regulatory approval and commercialization stages.
By delivering functional genes into patients’ cells, these therapies can address the underlying genetic cause of disease, making them a key component of precision medicine.
Dalnacogene Ponparvovec (BBM-H901)
Developer: Belief BioMed / Takeda
Approval Date: April 2025
Indication: Moderate-to-severe Hemophilia B
Clinical Value:
Dalnacogene ponparvovec uses an AAV vector to deliver the Factor IX gene, enabling patients to produce clotting protein continuously. This approach can significantly reduce or potentially replace long-term factor replacement therapy.
Future Trends of Approved Cell and Gene Therapy Drugs in China
China’s CGT industry is expanding rapidly, and more approved cell and gene therapy drugs in China are expected to emerge in the coming years.
1. Accelerating Technological Innovation
Many companies are developing next-generation therapies such as:
- CAR-NK therapies
- TCR-T therapies
- Gene-editing therapies
- AAV-based gene therapies
2. Rapid Growth of Clinical Research
China has become one of the most active regions for cell therapy clinical research, particularly in the field of CAR-T.
3. Improving Regulatory Framework
Regulatory authorities are gradually establishing more standardized oversight for advanced therapies, bringing CGT products into a more unified approval system.
4. Expanding Market Potential
Over the next decade, cell and gene therapies are expected to become one of the fastest-growing segments of the biopharmaceutical industry, with China playing an increasingly important role in the global CGT market.
Conclusion
Overall, the development of cell therapy vs gene therapy in China reflects the rapid progress of advanced therapeutics and the growing role of precision medicine in modern healthcare.
Cell therapy and gene therapy represent major advancements in modern medicine.
Cell therapy works primarily by modifying or infusing living cells to exert therapeutic effects, with CAR-T therapy currently being the most mature application. Gene therapy, on the other hand, treats disease by modifying or supplementing genetic information, making it particularly suitable for genetic and rare diseases.
💊 In China, as regulatory policies continue to evolve and technological breakthroughs accelerate, more innovative therapies are expected to receive approval and become approved cell and gene therapy drugs in China.
China pharmaceutical wholesaler DengYueMed continues to monitor developments in cell therapy, gene therapy, and other advanced therapeutic technologies, working to improve global patient access to innovative medicines.
FAQ about Approved Cell and Gene Therapy Drugs in China
What are approved cell and gene therapy drugs in China?
Approved cell and gene therapy drugs in China include CAR-T therapies, stem cell therapies, and gene therapies authorized by the National Medical Products Administration (NMPA). Currently, nine CGT products have been approved.
Which country is best for gene therapy?
There is no single country that is “best” for gene therapy. However, the United States currently leads in approved gene therapy products and clinical research, while China and United Kingdom are also major centers for gene therapy development and clinical trials.
Is stem cell therapy legal in China?
Yes, stem cell therapy is legal in China, but it is strictly regulated. Only therapies approved by the National Medical Products Administration or conducted within authorized clinical research programs are permitted.
What is cell therapy used for?
Cell therapy is used to treat diseases by introducing living cells that can repair tissues, regulate the immune system, or destroy cancer cells. It is commonly applied in conditions such as cancer, blood disorders, and some immune-related diseases.



