ADC Drugs by Target in 2026: Full List of 21 Approved Antibody-Drug Conjugates
Antibody-drug conjugates (ADCs) are one of the fastest-growing classes of therapies in precision oncology. In this guide to ADC drugs by target in 2026, we provide a comprehensive overview of approved antibody-drug conjugates and their classification based on tumor targets.
By combining highly potent cytotoxic drugs with monoclonal antibodies, ADCs enable “targeted delivery,” directing drugs specifically to tumor cells while minimizing damage to normal tissues.
With advances in antibody engineering, linker technology, and payload design, ADCs have entered rapid development, with growing clinical efficacy and global approvals.
🚀 As of March 2026, approximately 21 ADC drugs have been approved worldwide, targeting multiple tumor antigens such as HER2, TROP2, CD antigens, EGFR, BCMA, and Nectin-4. These ADC drugs play critical roles in the treatment of breast cancer, lung cancer, lymphoma, urothelial carcinoma, ovarian cancer, and multiple myeloma.
DengYueMed, a global pharmaceutical distributor, provides a systematic overview of these therapies through a structured analysis of ADC drugs by target in 2026.
I. Overview of ADC Drugs
This overview provides a structured understanding of adc drugs by target in 2026, highlighting how different targets correspond to specific therapies.
Their core structure includes:
1. Monoclonal Antibody (Antibody)
Responsible for recognizing specific antigens on tumor cell surfaces, enabling precise targeting.
2. Linker
Connects the antibody to the payload, remaining stable in circulation and releasing the drug inside tumor cells.
3. Cytotoxic Payload (Payload)
Typically consists of highly potent microtubule inhibitors or DNA-damaging agents that kill cancer cells.
The mechanism of action includes:
- The antibody binds to a tumor-specific antigen
- The ADC is internalized into tumor cells
- The linker is cleaved
- The payload is released, damaging DNA or microtubules and inducing cell death
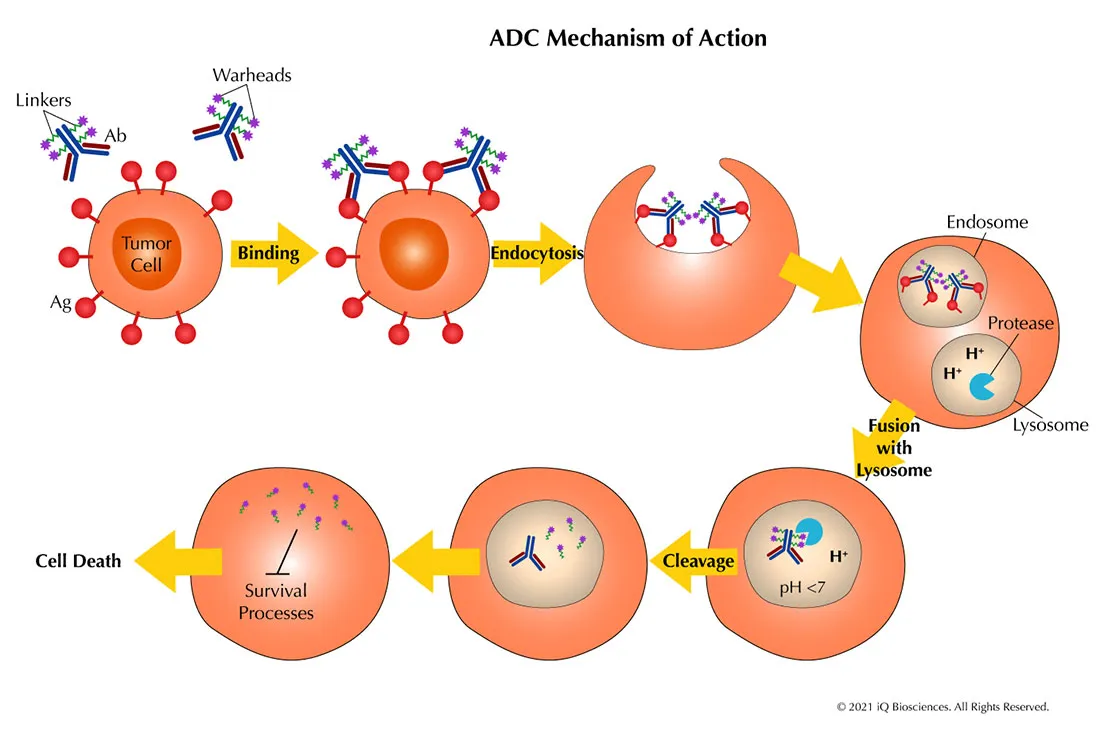
This mechanism underpins the classification of adc drugs by target in 2026, combining the advantages of targeted therapy and chemotherapy.
II. Global List of Approved ADC Drugs
As of March 2026, approximately 21 ADC drugs have been approved by the U.S. Food and Drug Administration (FDA) and other global regulatory authorities.
Key details are summarized below:
| Drug | Brand Name | Company | First Approval | Target | Main Indication |
|---|---|---|---|---|---|
| Gemtuzumab ozogamicin | Mylotarg | Pfizer | 2000 | CD33 | Acute Myeloid Leukemia |
| Brentuximab vedotin | Adcetris | Seagen/Takeda | 2011 | CD30 | Hodgkin Lymphoma |
| Trastuzumab emtansine | Kadcyla | Roche | 2013 | HER2 | HER2+ Breast Cancer |
| Inotuzumab ozogamicin | Besponsa | Pfizer | 2017 | CD22 | Acute Lymphoblastic Leukemia |
| Moxetumomab pasudotox | Lumoxiti | AstraZeneca / MedImmune | 2018 | CD22 | Hairy Cell Leukemia |
| Polatuzumab vedotin | Polivy | Roche | 2019 | CD79b | DLBCL |
| Enfortumab vedotin | Padcev | Astellas/Seagen | 2019 | Nectin-4 | Urothelial Cancer |
| Trastuzumab deruxtecan | Enhertu | Daiichi Sankyo | 2019 | HER2 | Breast Cancer, Lung Cancer |
| Sacituzumab govitecan | Trodelvy | Gilead | 2020 | TROP2 | TNBC |
| Belantamab mafodotin | Blenrep | GSK | 2020 | BCMA | Multiple Myeloma |
| Cetuximab sarotalocan | Akalux | Rakuten | 2020 | EGFR | Head and Neck Cancer |
| Loncastuximab tesirine | Zynlonta | ADC Therapeutics | 2021 | CD19 | Lymphoma |
| Disitamab vedotin | Aidixi | RemeGen | 2021 | HER2 | Gastric Cancer |
| Tisotumab vedotin | Tivdak | Seagen | 2021 | Tissue Factor | Cervical Cancer |
| Mirvetuximab soravtansine | Elahere | ImmunoGen | 2022 | FRα | Ovarian Cancer |
| Sacituzumab tirumotecan | Jiataile | Kelun-Biotech | 2024 | TROP2 | TNBC, NSCLC |
| Datopotamab deruxtecan | Datroway | AstraZeneca | 2024 | TROP2 | Breast Cancer |
| Telisotuzumab vedotin | Emrelis | AbbVie | 2025 | c-MET | NSCLC |
| Trastuzumab rezetecan | Aiweida | Hengrui | 2025 | HER2 | NSCLC |
| Trastuzumab botidotin | Shutailai | Kelun | 2025 | HER2 | Breast Cancer |
| Becotatug vedotin | Meiyouheng | Lepu | 2025 | EGFR | Nasopharyngeal Cancer |
III. ADC Drugs by Target in 2026 (Target-Based Classification)
1. HER2-Targeted ADC Drugs
HER2 (Human Epidermal Growth Factor Receptor 2) is one of the most actively studied targets in ADC development. It belongs to the EGFR receptor family and plays a critical role in cell proliferation and signaling pathways.
In breast cancer, gastric cancer, and lung cancer, HER2 gene amplification or protein overexpression promotes tumor growth and progression, making it an important therapeutic target.
HER2-targeted ADCs use anti-HER2 monoclonal antibodies to precisely recognize tumor cells and deliver cytotoxic payloads into cancer cells.
Representative drugs include:
● Trastuzumab emtansine (Kadcyla)
● Trastuzumab deruxtecan (Enhertu)
● Disitamab vedotin (Aidixi)
● Trastuzumab rezetecan (Aiweida)
● Trastuzumab botidotin (Shutailai)
Among them, Enhertu is considered one of the most successful ADC drugs. Its DXd payload has a strong bystander effect, enabling efficacy even in HER2-low tumors.
2. TROP2-Targeted ADC Drugs
TROP2 (Trophoblast Cell Surface Antigen 2) is a transmembrane glycoprotein highly expressed in various epithelial tumors, including breast, lung, pancreatic, and colorectal cancers.
TROP2 is involved in tumor proliferation, migration, and invasion, making it a major focus of ADC development.
TROP2-targeted ADCs typically carry topoisomerase inhibitors and deliver them selectively to tumor cells.
Representative drugs:
● Sacituzumab govitecan (Trodelvy)
● Sacituzumab tirumotecan (Jiataile)
● Datopotamab deruxtecan (Datroway)
These drugs are mainly used in triple-negative breast cancer (TNBC), HR+/HER2- breast cancer, and non-small cell lung cancer.
Trodelvy was the first approved TROP2 ADC, delivering SN-38 as its payload. With the emergence of DXd-based ADCs, TROP2-targeted therapies are becoming increasingly important.
3. CD-Targeted ADC Drugs
CD antigens are widely expressed on immune cells and are key therapeutic targets in hematologic malignancies.
These targets are highly stable and specific, making them ideal for ADC-based therapies.
CD-targeted ADCs bind to leukemia or lymphoma cells and release potent cytotoxic agents.
Representative drugs include:
● Gemtuzumab ozogamicin (CD33)
● Brentuximab vedotin (CD30)
● Inotuzumab ozogamicin (CD22)
● Loncastuximab tesirine (CD19)
● Polatuzumab vedotin (CD79b)
These drugs are used in:
- Acute Myeloid Leukemia (AML)
- Hodgkin Lymphoma
- Diffuse Large B-Cell Lymphoma (DLBCL)
- Acute Lymphoblastic Leukemia (ALL)
Brentuximab vedotin is a landmark CD30-targeted ADC with significant clinical impact.
4. EGFR-Targeted ADC Drugs
EGFR is a key oncogenic driver in many solid tumors, including head and neck cancer, nasopharyngeal carcinoma, and some lung cancers.
It regulates cell proliferation, differentiation, and angiogenesis.
Recent research has focused on EGFR-targeted ADCs as an alternative to traditional EGFR inhibitors.
Relevant ADC drugs:
- Cetuximab sarotalocan (Akalux)
- Becotatug vedotin (Meiyouheng)
Akalux uses an innovative Photoimmunotherapy (PIT) approach. After binding tumor cells, it is activated by near-infrared light, causing membrane disruption and tumor cell death.
5. Tissue Factor-Targeted ADC Drugs
Tissue Factor (TF) is a transmembrane protein involved in the coagulation cascade and is highly expressed in multiple solid tumors, including cervical, ovarian, and pancreatic cancers.
TF also contributes to tumor angiogenesis and metastasis.
Representative drug:
Tisotumab vedotin (Tivdak)
This is the first ADC approved for recurrent or metastatic cervical cancer. It delivers MMAE payload and has shown strong response rates in clinical trials.
6. BCMA-Targeted ADC Drugs
BCMA (B-cell maturation antigen) is highly expressed on multiple myeloma cells.
It is also widely used in CAR-T and bispecific antibody development.
Representative drug:
Belantamab mafodotin (Blenrep)
It delivers MMAF payload to myeloma cells. Although withdrawn in some markets, BCMA remains a highly promising target.
7. Nectin-4-Targeted ADC Drugs
Nectin-4 is a cell adhesion molecule highly expressed in urothelial carcinoma, with expression rates exceeding 90% in advanced bladder cancer.
Representative drug:
Enfortumab vedotin (Padcev)
Padcev delivers MMAE payload and has become a standard treatment. It is also used in combination with pembrolizumab (Keytruda).
8. FRα-Targeted ADC Drugs
FRα (Folate Receptor Alpha) is highly expressed in ovarian cancer and minimally expressed in normal tissues.
Representative drug:
Mirvetuximab soravtansine (Elahere)
It delivers DM4 payload and provides a targeted treatment option for platinum-resistant ovarian cancer.
9. c-MET-Targeted ADC Drugs
c-MET is a receptor tyrosine kinase involved in tumor growth, invasion, and metastasis.
It is overexpressed in some NSCLC patients.
Representative drug:
Telisotuzumab vedotin (Emrelis)
This ADC delivers MMAE payload and has shown promising response rates in c-MET overexpressing NSCLC.
IV. ADC Technology Development Trends
With ongoing innovation, ADCs are rapidly evolving toward improved precision, efficacy, and safety. Recent studies published in Nature Reviews Drug Discovery highlight rapid advances in ADC design and payload optimization.
In the era of adc drugs by target in 2026, technological advances are expanding their role across multiple tumor types.
Next-generation ADCs are evolving toward:
- More stable linker technology
Enhances stability in circulation and reduces premature payload release, improving safety. - More potent payloads (e.g., DXd)
Enables stronger antitumor activity and effectiveness in low-expression tumors. - Higher drug-to-antibody ratio (DAR)
Increases payload delivery while requiring optimized balance for safety and stability. - Stronger bystander effects
Allows killing of neighboring tumor cells, especially in heterogeneous solid tumors. - Broader target exploration
New targets such as TROP2 and Nectin-4 are expanding the landscape of ADC drugs.
These advancements are driving ADCs to become a cornerstone of modern cancer therapy.
Conclusion
Since the first ADC drug was approved in 2000, the field has undergone more than two decades of rapid innovation.
As of March 2026, 21 ADC drugs have been approved globally, covering multiple tumor targets and indications.
Understanding adc drugs by target in 2026 is essential for researchers, clinicians, and pharmaceutical partners.
✨ As a global distributor focusing on innovative oncology therapies, DengYueMed continues to track the development of ADC technologies and provide reliable drug information and supply support for international partners.
FAQ about ADC Drugs by Target in 2026
What are the targets of ADC drugs?
ADC drugs target specific antigens expressed on the surface of cancer cells. Common targets include HER2, TROP2, CD antigens, and BCMA, which enable precise delivery of cytotoxic agents to tumor cells.
What are the top ADC drugs?
Top ADC drugs are typically those with strong clinical efficacy and broad use across cancers. Leading examples include trastuzumab deruxtecan (Enhertu), trastuzumab emtansine (Kadcyla), sacituzumab govitecan (Trodelvy), enfortumab vedotin (Padcev), and brentuximab vedotin (Adcetris).
Which ADCs are FDA approved?
More than 10 ADCs are FDA approved for cancer treatment. Examples include trastuzumab deruxtecan (Enhertu), enfortumab vedotin (Padcev), and sacituzumab govitecan (Trodelvy), covering both solid tumors and hematologic cancers.
Is ADC a type of targeted therapy?
Yes, ADCs (antibody-drug conjugates) are a type of targeted therapy. They use antibodies to specifically bind to tumor-associated antigens and deliver cytotoxic drugs directly into cancer cells, improving precision and reducing damage to normal tissues.