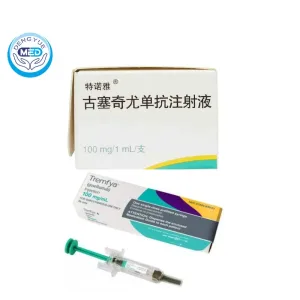
Tremfya (Guselkumab) | Plaque Psoriasis | DengYue Medicine
- Generic Name/Brand Name: Guselkumab/Tremfya
- Indications: Plaque psoriasis, active psoriatic arthritis, and ulcerative colitis
- Dosage Form: Injection for subcutaneous use
- Specification: 100 mg/mL x 1 prefilled syringe
Guselkumab Application Scope
Moderate to severe plaque psoriasis, active psoriatic arthritis, and moderate to severe ulcerative colitis in adults.
Characteristics
-
Ingredients:
-
Active Ingredient: Guselkumab
-
Excipients:
-
Subcutaneous Injection (Prefilled Syringe/Pen): L-histidine, L-histidine monohydrochloride monohydrate, polysorbate 80, sucrose, water for injection.
-
Intravenous Infusion (Vial): EDTA disodium dihydrate, L-histidine, L-histidine monohydrochloride monohydrate, L-methionine, polysorbate 80, sucrose, water for injection.
-
-
-
Properties:
-
Type: Monoclonal antibody
-
Mechanism of Action: Selective inhibitor of interleukin-23 (IL-23), specifically targeting the p19 subunit, thereby inhibiting the activation of T-helper cells involved in inflammatory processes.
-
-
Packaging Specification:
-
Subcutaneous Injection:
-
200 mg/2 mL (100 mg/mL) in a single-dose prefilled syringe.
-
200 mg/2 mL (100 mg/mL) in a single-dose prefilled pen.
-
-
Intravenous Infusion:
-
200 mg/20 mL (10 mg/mL) solution in a single-dose vial.
-
-
-
Storage:
-
Subcutaneous Injection:
-
Refrigerate at 2°C to 8°C (36°F to 46°F).
-
May be stored at room temperature (up to 25°C or 77°F) for a maximum of 30 days.
-
-
Intravenous Infusion:
-
Refrigerate at 2°C to 8°C (36°F to 46°F).
-
Do not freeze.
-
Protect from light.
-
-
-
Expiry Date: Refer to the expiration date printed on the packaging.
-
Executive Standard: Approved by regulatory authorities such as the U.S. FDA and EMA.
-
Approval Number: U.S. FDA Biologics License Application (BLA) 761061.
-
Date of Revision: March 2025.
-
Manufacturer: Janssen Biotech, Inc., a subsidiary of Johnson & Johnson.
Guidelines for the Use of Guselkumab
-
Dosage and Administration:
-
Plaque Psoriasis:
-
Induction: 100 mg subcutaneously at weeks 0, 4.
-
Maintenance: 100 mg subcutaneously every 8 weeks.
-
-
Psoriatic Arthritis:
-
Induction: 100 mg subcutaneously at weeks 0, 4.
-
Maintenance: 100 mg subcutaneously every 8 weeks.
-
May be used alone or in combination with conventional disease-modifying antirheumatic drugs (cDMARDs) such as methotrexate.
-
-
Ulcerative Colitis:
-
Induction: 200 mg subcutaneously at weeks 0, 4.
-
Maintenance: 200 mg subcutaneously every 8 weeks.
-
-
Administration:
-
Subcutaneous injections should be administered by a healthcare professional or a trained patient/caregiver.
-
Allow the prefilled syringe or pen to reach room temperature before injection.
-
Inspect the solution for particulate matter or discoloration before use; do not use if discolored or contains particles.
-
Rotate injection sites to minimize tissue irritation.
-
-
-
Adverse Reactions:
-
Common (≥10%):
-
Upper respiratory tract infections.
-
-
Less Common (1–10%):
-
Headache.
-
Injection site reactions.
-
Arthralgia.
-
Elevated liver enzymes.
-
Diarrhea.
-
Gastroenteritis.
-
Tinea infections.
-
Herpes simplex infections.
-
-
Serious:
-
Hypersensitivity reactions, including anaphylaxis.
-
Serious infections.
-
-
-
Contraindications: History of serious hypersensitivity reactions to guselkumab or any of its excipients.
-
Precautions:
-
Monitor for signs of infection; discontinue if a serious infection develops.
-
Evaluate for tuberculosis before initiating therapy.
-
Use with caution in patients with a history of recurrent infections.
-
Not recommended for use during pregnancy unless clearly needed.
-
Lactation: Unknown if distributed in human milk; maternal IgG is known to be present in human milk.
-
Interactions
- Drug Interactions:
-
No known significant drug interactions.
-
Caution when used concomitantly with other immunosuppressive agents due to increased risk of infections.
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.


.webp)




Reviews
There are no reviews yet.