Agalsidase Alfa | Anti Fabry Disease
- Generic Name/Brand Name: Agalsidase Alfa/Replagal
- Indications: Agalsidase Alfa is a new and special drug indicated for Fabry disease and glycogen accumulation disease (type I, type II)
- Dosage Form: Injection
- Specification: 3.5 mg (3.5 mL)/vial
Agalsidase Alfa Application Scope
Agalsidase Alfa is used for the treatment of Fabry disease (α-galactosidase A deficiency), as a new and special drug and a long-term enzyme replacement therapy. It is suitable for adults, children, and adolescents. The safety and efficacy for children aged 0–6 years have not been established.
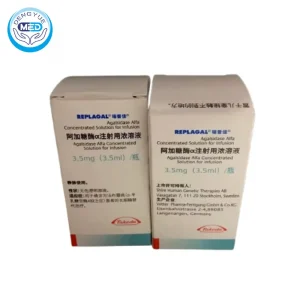
Agalsidase Alfa Characteristics
- Ingredients: Concentrated solution for injection
- Properties: Treats α-galactosidase A deficiency
- Specification: 3.5 mg (3.5 mL)/vial
- Packaging Specification: Each box contains 1, 4, or 10 vials of 3.5 mL.
- Storage: 2°C to 8°C (36°F to 46°F). Do not freeze. Avoid light.
- Expiry Date: 24months
- Executive Standard: JX20200106
- Approval Number: SJ20200021
- Date of Revision: According to the latest drug insert.
- Manufacturer: Shire Human Genetic Therapies AB
Guidelines For The Use Of Agalsidase Alfa
Medication Limitations
Agalsidase Alfa Interactions
Interactions of Agalsidase with other drugs have not been determined. Before starting treatment, inform your doctor of all medications you are using.
Note: If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYueMed. It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.




Reviews
There are no reviews yet.