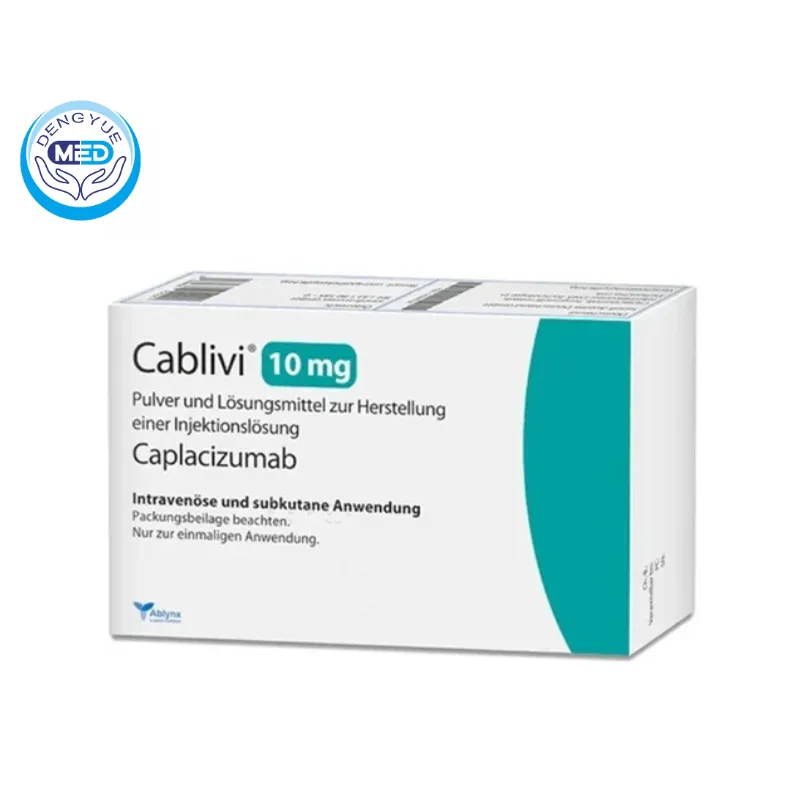
Caplacizumab|Thrombotic Thrombocytopenic Purpura|HongKong DengYue Medicine
- Generic Name/Brand Name: Caplacizumab/Cablivi
- Indications: Thrombotic Thrombocytopenic Purpura
- Dosage Form: Lyophilized powder for injection
- Specification: 11 mg per vial
Caplacizumab Application Scope
Caplacizumab is indicated for the treatment of adults experiencing an episode of acquired thrombotic thrombocytopenic purpura (aTTP), in combination with plasma exchange and immunosuppressive therapy.
Caplacizumab Characteristics
Ingredients:
-
Active Ingredient: Caplacizumab-yhdp
-
Excipients: Sucrose, citric acid monohydrate, sodium citrate dihydrate, polysorbate 80 .
Properties:
Caplacizumab is a humanized bivalent Nanobody® (single-domain antibody) that targets von Willebrand factor (vWF), inhibiting the interaction between vWF and platelets, thereby preventing platelet adhesion and aggregation .
Specification:
-
Dosage Form: Lyophilized powder for injection
-
Strength: 11 mg per vial .
Packaging Specification:
Each package contains:
-
1 vial of Caplacizumab 11 mg
-
1 prefilled syringe with 1 mL sterile water for injection
-
1 vial adapter
-
1 needle .
Storage:
-
Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light.
-
Do not freeze.
-
Unopened vials may be stored at room temperature up to 30°C (86°F) for a single period of up to 2 months. Do not return to the refrigerator after storage at room temperature .
Expiry Date:
-
48 months from the date of manufacture when stored at 2°C to 8°C .
Executive Standard:
-
U.S. FDA Biologics License Application (BLA) No. 761112 .
Approval Number:
-
BLA 761112 .
Date of Revision:
-
February 2019 .
Manufacturer:
-
Ablynx, a Sanofi company .
Guidelines for the Use of Caplacizumab
Dosage and Administration:
-
Initial Dose: 11 mg intravenous bolus injection prior to the first plasma exchange session.
-
Subsequent Doses: 11 mg subcutaneous injection once daily after each plasma exchange session.
-
Treatment Duration: Continue daily subcutaneous injections for 30 days after the last plasma exchange session. If signs of persistent underlying disease are present, treatment may be extended for up to 28 additional days .
Adverse Reactions:
Common side effects include:
-
Epistaxis (nosebleeds)
-
Headache
-
Gingival bleeding
-
Fatigue
-
Fever
-
Injection site reactions (bleeding, itching)
-
Back pain
-
Muscle pain
-
Numbness and tingling
-
Urinary tract infection
-
Blood in urine
-
Abnormal vaginal bleeding
-
Shortness of breath
-
Hives .
Contraindications:
-
Severe hypersensitivity reaction to Caplacizumab-yhdp or any of its excipients .
Precautions:
-
Increased risk of bleeding: Use with caution in patients with underlying bleeding disorders or those on anticoagulant or antiplatelet therapy. Monitor for signs of bleeding .
Caplacizumab Interactions
Drug Interactions:
-
Concomitant use with anticoagulants (e.g., warfarin, heparin) or antiplatelet agents (e.g., aspirin, clopidogrel) may increase the risk of bleeding. Avoid concurrent use when possible. If used together, monitor closely for bleeding .
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.



Reviews
There are no reviews yet.