Brukinsa (Zanubrutinib) – CLL | DengYueMed
- Generic Name/Brand Name: Zanubrutinib/Brukinsa
- Indications: Chronic Lymphocytic Leukemia (Hematology)
- Dosage Form: Capsule
- Specification: 80 mg*64 capsules
Brukinsa Application Scope
Brukinsa is an oral, selective Bruton’s tyrosine kinase (BTK) inhibitor used to treat several B-cell malignancies, including CLL/SLL, MCL, WM, MZL, and FL.
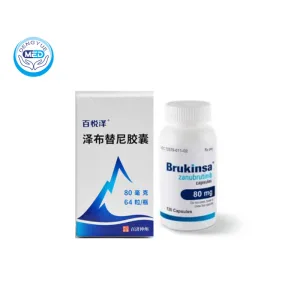
Characteristics
-
Ingredients: Zanubrutinib
-
Properties: Small oral capsule; BTK inhibitor targeting B-cell receptor signaling
-
Packaging Specification: Capsules, 80 mg gelatin‑coated, white/off‑white, marked “ZANU 80”
-
Storage: Store at controlled room temperature; Refer to packaging for precise conditions
-
Expiry Date: Refer to the printed label on each pack
-
Executive Standard: Follows FDA and EMA product monographs
-
Approval Number:
-
FDA approval for MCL Nov 2019
-
Expanded indications approved in January 2023 for CLL/SLL
-
-
Date of Revision: US prescribing info updated Jan 2025; Canadian monograph revised Feb 17, 2022
-
Manufacturer: BeiGene (BeiGene Switzerland GmbH, Basel)
Guidelines for the Use of Brukinsa
-
Dosage and Administration:
-
160 mg orally twice daily or 320 mg once daily until disease progression/toxicity
-
Take with or without food; swallow capsules whole with water
-
If missed, take same day; resume usual schedule next day
-
For severe hepatic impairment (Child‑Pugh C): 80 mg twice daily
-
-
Adverse Reactions:
-
Common (≥30 %):
-
decreased neutrophils, thrombocytopenia
-
upper respiratory infection, bleeding, musculoskeletal pain
-
-
Serious risks:
-
hemorrhage, infections, cytopenias, cardiac arrhythmias
-
liver injury, second primary malignancies, embryo‑fetal toxicity
-
-
-
Contraindications: Known allergy to zanubrutinib or any capsule excipients
-
Precautions:
-
Monitor blood counts, liver function, signs of bleeding/infection/arrhythmia
-
Avoid use around surgery; assess bleeding risk when on anticoagulants
-
Use contraception during and after treatment (women ≥1 week; men ≥3 months)
-
Avoid breastfeeding while taking and for 2 weeks after the last dose
-
Brukinsa Interactions
-
Drug Interactions:
-
Metabolized by CYP3A. Strong/moderate inhibitors require dose reduction
-
Strong/moderate inducers should be avoided, or the dosage increased to 320 mg BID
-
Anticoagulants/antiplatelet agents may increase bleeding risk
-
Note:
- If there is a new packaging for the drug, the new packaging shall prevail. The above information is sourced from DengYue Medicine.
- It is only for internal discussion among medical staff and does not serve as a basis for medication. For specific medication guidelines, please consult the attending physician.





Reviews
There are no reviews yet.